| In vitro: |
| Daru. 2013 Jan 2;21(1):2. | | Urease inhibitory activities of β-boswellic acid derivatives.[Pubmed: 23351363 ] | Boswellia carterii have been used in traditional medicine for many years for management different gastrointestinal disorders. In this study, we wish to report urease inhibitory activity of four isolated compound of boswellic acid derivative.
METHODS AND RESULTS:
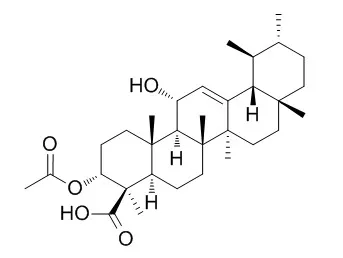
4 pentacyclic triterpenoid acids were isolated from Boswellia carterii and identified by NMR and Mass spectroscopic analysis (compounds 1, 3-O-acetyl-9,11-dehydro-β-boswellic acid; 2, 3-O-Acetyl-11-hydroxy-beta-boswellic acid ; 3. 3-O- acetyl-11-keto-β-boswellic acid and 4, 11-keto-β-boswellic acid. Their inhibitory activity on Jack bean urease were evaluated. Docking and pharmacophore analysis using AutoDock 4.2 and Ligandscout 3.03 programs were also performed to explain possible mechanism of interaction between isolated compounds and urease enzyme. It was found that compound 1 has the strongest inhibitory activity against Jack bean urease (IC50 = 6.27 ± 0.03 μM), compared with thiourea as a standard inhibitor (IC50 = 21.1 ± 0.3 μM).
CONCLUSIONS:
The inhibition potency is probably due to the formation of appropriate hydrogen bonds and hydrophobic interactions between the investigated compounds and urease enzyme active site and confirms its traditional usage. | | J Nat Prod. 2000 Aug;63(8):1058-61. | | Workup-dependent formation of 5-lipoxygenase inhibitory boswellic acid analogues.[Pubmed: 10978197] | Pentacyclic triterpenes from the 11-keto-boswellic acid series were identified as the active principal ingredients of Boswellia resin, inhibiting the key enzyme of leukotriene biosynthesis, 5-lipoxygenase (5-LO).
Of the genuine boswellic acids hitherto characterized, 3-O-acetyl-11-keto-beta-boswellic acid, AKBA (1), proved to be the most potent inhibitor of 5-LO.
METHODS AND RESULTS:
In the course of purification of further boswellic acid derivatives from Boswellia resin, we observed the degradation of the natural compound 3-O-Acetyl-11-hydroxy-beta-boswellic acid (2) to the thermodynamically more stable product 3-O-acetyl-9, 11-dehydro-beta-boswellic acid (4). The metastable intermediate of this conversion, under moderate conditions of workup in methanolic solutions, was identified as 3-O-acetyl-11-methoxy-beta-boswellic acid (3). The novel artifactual boswellic acid derivatives inhibited 5-LO product formation in intact cells with different characteristics: 4 almost totally abolished 5-LO activity, with an IC(50) of 0.75 microM, whereas 3 and 9,11-dehydro-beta-boswellic acid (5), the deacetylated analogue of 4, were incomplete inhibitors.
CONCLUSIONS:
The data suggest that the conditions chosen for the workup of Boswellia extracts could significantly influence the potency of their biological actions and their potential therapeutic effectiveness. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)