| In vitro: |
| Food Funct. 2013 Nov;4(11):1632-8. | | Isolation and quantification of major chlorogenic acids in three major instant coffee brands and their potential effects on H2O2-induced mitochondrial membrane depolarization and apoptosis in PC-12 cells.[Pubmed: 24061869] | Coffee is a most consumed drink worldwide, with potential health effects on several chronic diseases including neuronal degenerative diseases. Chlorogenic acids (CHAs) are phenolic compounds found in coffee and they are reported to have strong antioxidant and anti-inflammatory activities. However, the amounts of CHAs often vary in coffee drinks and their potential effects on ROS-induced neuronal cell death still require more investigation.
METHODS AND RESULTS:
Therefore, in this paper, major CHAs were isolated from three major instant coffee brands, confirmed and quantified using HPLC and NMR spectroscopic methods. Then, their antioxidant activities and protective effects on H2O2-induced apoptosis in PC-12 cells were investigated using radical scavenging, mitochondrial membrane potential and caspase assays. In the coffee samples, three major CHAs (3-O-caffeoylquinic acid, 4-O-caffeoylquinic acid, 5-O-caffeoylquinic acid) and some minor CHAs (3-O-Feruloylquinic acid, 4-O-feruloylquinic acid, 5-O-feruloylquinic acid, 3,5-O-dicaffeoylquinic acid, 3,4-O-dicaffeoylquinic acid, and 4,5-O-dicaffeoylquinic acid) were detected. The three major CHAs were further isolated and their chemical structures were confirmed using NMR spectroscopic techniques. Also, the amounts of the three major CHAs were individually quantified using a HPLC method. At the concentration of 10 μM, all three major CHAs quenched DPPH and/or xanthine oxidase-generated radical species by 21-51% (P < 0.014). They also inhibited H2O2-induced mitochondrial membrane depolarization and caspase-9 activation by 27% (P < 0.034) and 50% (P < 0.05), respectively.
CONCLUSIONS:
This study suggests that the major CHAs found in coffee are likely to be potent antioxidant compounds able to quench radical species as well as inhibit H2O2-induced apoptosis via suppressing mitochondrial membrane depolarization and caspase-9 activation in the cells. | | Yao Xue Xue Bao. 2015 Feb;50(2):207-10. | | Chemical constituents of the roots of Macleaya microcarpa and activation efficacy of benzophenanthridine alkaloids for the transcription of xbp1 gene.[Pubmed: 25975030] | Ongoing study on the chemical constituents of the roots of Macleaya microcarpa led to the isolation of eight compounds of derivatives of triterpenes and organic acids in addition to some previously identified benzophenanthridines.
METHODS AND RESULTS:
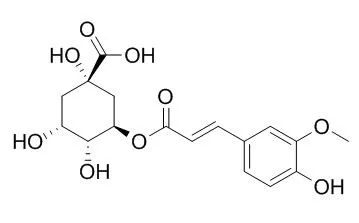
The eight compounds were identified by spectroscopic methods as well as comparison with literature values as 1-oxo-2, 22 (30)-hopandien-29-oic acid (1), 3-oxo-12-oleanen-30-oic acid (2), 3α-hydroxy-12-oleanen-30-oic acid (3), 3β-hydroxy-12-oleanen-30-oic acid (4), ferulic acid (5), ferulic acid 4-O-β-D-glucoside (6), 3-O-Feruloylquinic acid (7), and methyl 3-O-feruloylquinate (8). Of which, 1 is a new triterpenoid of hopanes and 2-8 are isolated from M microcarpa for the first time. In order to discover natural active compounds as potential agents of anti-ulcerative colitis (UC), an in vitro drug high-throughput screening model targeted x-box-binding protein 1 (xbp1) was employed to evaluate the activity of the major chemical constituents of M microcarpa.
CONCLUSIONS:
The result confirmed that two dihydrobenzophenanthridines, dihydrosanguinarine (9) and dihydrochelerythrine (10), showed a certain activity on activating the transcription of xbpl, a transcription factor (TF) associated with the occurrence, development, and potential treatment of UC, with their relative activating ratios being 1.76 and 1.77 times, respectively, as compared with control group. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)