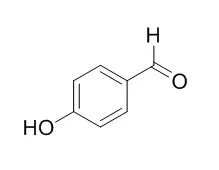
ChemFaces products have been cited in many studies from excellent and top scientific journals
Contact Us
Order & Inquiry & Tech Support
Tel: (0086)-27-84237683
Tech: service@chemfaces.com
Order: manager@chemfaces.com
Address: 176, CheCheng Eest Rd., WETDZ, Wuhan, Hubei 430056, PRC
How to Order
Orders via your E-mail:
1. Product number / Name / CAS No.
2. Delivery address
3. Ordering/billing address
4. Contact information
Order: manager@chemfaces.com
Delivery time
Delivery & Payment method
1. Usually delivery time: Next day delivery by 9:00 a.m. Order now
2. We accept: Wire transfer & Credit card & Paypal
Citing Use of our Products
* Packaging according to customer requirements(5mg, 10mg, 20mg and more). We shipped via FedEx, DHL, UPS, EMS and others courier.
According to end customer requirements, ChemFaces provide solvent format. This solvent format of product intended use: Signaling Inhibitors, Biological activities or Pharmacological activities.
| Size /Price /Stock |
10 mM * 1 mL in DMSO / $7.0 / In-stock |
Other Packaging |
*Packaging according to customer requirements(100uL/well, 200uL/well and more), and Container use Storage Tube With Screw Cap |
More articles cited ChemFaces products.
- Int Immunopharmacol.2019, 71:361-371
- Applied Biological Chemistry...2022...
- Biomol Ther (Seoul).2019, 10.4062
- Planta Med.2018, 84(6-07):465-474
- Phytomedicine.2015, 22(11):1027-36
- Front Microbiol.2021, 12:736780.
- J Food Sci Technol.2022, 59(1):212-219.
- Anticancer Res.2018, 38(4):2127-2135
- Pest Manag Sci.2023, 79(8):2675-2685.
- Microbiol. Biotechnol. Lett....2022...
- Nutrients.2022, 14(19):4170.
- Molecules.2019, 24(20):3755
- Asian Pac J Cancer Prev. ...2020...
- Ind Crops Prod.2015, 67:185-191
- Front Pharmacol.2016, 7:460
- J Nat Sc Biol Med2019, 10(2):149-156
- J Ethnopharmacol.2017, 206:73-77
- JEJU National University2022, 24032.
- Molecules.2019, 24(24):E4536
- Pharmaceutics.2020, 12(9):882.
- The Journal of Korean Medicine...2022...
- Oncotarget.2016, 8(51):88386-88400
- Cancers (Basel).2021, 13(17):4327.
- More...
Our products had been exported to the following research institutions and universities, And still growing.
- Universidad de Buenos Aires (Argentina)
- Universidade do Porto (Portugal)
- Celltrion Chemical Research Ins... (Korea)
- Chungnam National University (Korea)
- Institute of Bioorganic Chemist... (Poland)
- Shanghai Institute of Organic C... (China)
- University of Toronto (Canada)
- Universidade de Franca (Brazil)
- Molecular Biology Institute of ... (Spain)
- Colorado State University (USA)
- University of Indonesia (Indonesia)
- More...






 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)