| In vitro: |
| Strahlenther Onkol. 2015 May;191(5):429-36. | | 8-prenylnaringenin and tamoxifen inhibit the shedding of irradiated epithelial cells and increase the latency period of radiation-induced oral mucositis : cell culture and murine model.[Pubmed: 25432325] | The major component in the pathogenesis of oral radiation-induced mucositis is progressive epithelial hypoplasia and eventual ulceration. Irradiation inhibits cell proliferation, while cell loss at the surface continues. We conceived to slow down this desquamation by increasing intercellular adhesion, regulated by the E-cadherin/catenin complex. We investigated if 8-Prenylnaringenin (8-PN) or tamoxifen (TAM) decrease the shedding of irradiated human buccal epithelial cells in vitro and thus delay the ulcerative phase of radiation-induced mucositis in vivo.
METHODS AND RESULTS:
In vitro, aggregates of buccal epithelial cells were irradiated and cultured in suspension for 11 days. 8-PN or TAM were investigated regarding their effect on cell shedding. In vivo, the lower tongue surface of mice was irradiated with graded single doses of 25 kV X-rays. The incidence, latency, and duration of the resulting mucosal ulcerations were analyzed after topical treatment with 8-PN, TAM or solvent.
8-PN or TAM prevented the volume reduction of the irradiated cell aggregates during the incubation period. This was the result of a higher residual cell number in the treated versus the untreated irradiated aggregates. In vivo, topical treatment with 8-PN or TAM significantly increased the latency of mucositis from 10.9 to 12.1 and 12.4 days respectively, while the ulcer incidence was unchanged.
CONCLUSIONS:
8-PN and TAM prevent volume reduction of irradiated cell aggregates in suspension culture. In the tongues of mice, these compounds increase the latency period. This suggests a role for these compounds for the amelioration of radiation-induced mucositis in the treatment of head and neck tumors. | | Planta Med. 2015 Mar;81(4):305-11. | | Neurodifferentiating potential of 8-prenylnaringenin and related compounds in neural precursor cells and correlation with estrogen-like activity.[Pubmed: 25714726] | Neurodegenerative diseases are an increasing burden for our ageing societies; there is an as yet unmet need for the development of effective therapies. Neurogenesis, i.e., the generation of new neurons in the adult brain from neural stem cells, has received increasing attention since it offers the potential for endogenous brain repair and functional regeneration. Adult neurogenesis is partially under the control of sex hormones such as estradiol, and boosting neurogenesis with estradiol in animals correlates with cognitive improvement. 8-Prenylnaringenin imitates as highly potent phytoestrogen the effects of estradiol.
METHODS AND RESULTS:
Here, we studied the potential of 8-Prenylnaringenin, 6-prenylnaringenin, and related compounds on differentiation induction in vitro using neural precursor cells transiently transfected with a doublecortin promoter luciferase construct, which was recently shown to indicate neuronal fate and differentiation. The flavanones 8-Prenylnaringenin and 6-prenylnaringenin showed slight activity in this assay but significant activity by immunostaining. Although the estrogen-like activities of 8-Prenylnaringenin and 6-prenylnaringenin are very different, the activity in differentiation induction is similar. Interestingly, also some prenylflavonoids with extended prenyl groups, e.g., a geranyl group, showed increased differentiation activity, while estrogen-like activity is decreased.
CONCLUSIONS:
This allows the conclusion that estrogen-like activity of prenylflavanones does not correlate directly with the activity of differentiation induction in neural precursor cells. | | Life Sci . 2019 Sep 1;232:116633. | | Antiproliferative and apoptotic activities of 8-prenylnaringenin against human colon cancer cells[Pubmed: 31278947] | | Abstract
Aims: The compound 8-Prenylnaringenin (8-PN) is a prenylflavonoid that can be isolated from hops and beer and has anti-cancer properties against breast cancer. The aim of this study is to investigate the anti-proliferative and apoptotic activities of 8-PN against human colon cancer HCT-116 cells.
Main methods: Colon cancer HCT-116 cells were treated with 8-PN and subjected to MTT and acridine orange/propidium iodide (AO/PI) staining to investigate the cytotoxicity of 8-PN. Arrest of the cells at different phases of cell cycle was monitored in the presence of 8-PN. Moreover, the apoptotic effects of 8-PN was assessed via annexin V and caspase activity assays and compared to the untreated cells.
Key findings: The findings showed that 8-PN revealed strong inhibitory effect against HCT-116 cells with an IC50 value of 23.83 ± 2.9 μg/ml after 48 h. However, at similar concentrations and experimental time-points, the compound did not show cytotoxic effect to non-cancerous colon cells (CCD-41). Annexin-V assay indicates that 38.5% and 14.4% of HCT-116 cells had entered early and late stages of apoptosis, respectively after exposure of the cells to 8-PN for 48 h. Caspase activity assay illustrates that apoptosis is activated through both intrinsic and extrinsic pathways. Moreover, flow cytometry cell cycle results indicate that treatment with 8-PN significantly arrested the HCT-116 cells at G0/G1 phase.
Significance: These findings reveal that 8-PN has anti-proliferative activity against HCT-116 colon cancer cells via induction of intrinsic and extrinsic pathway-mediated apoptosis. Further investigations should be carried out to unravel the mechanistic pathways underlying these activities.
Keywords: 8-Prenylnaringenin; Anti-proliferation; Apoptosis; Caspases; Cell cycle arrest; Colon cancer. | | Molecules . 2017 Jun 30;22(7):1092. | | In Vitro Effect of 8-Prenylnaringenin and Naringenin on Fibroblasts and Glioblastoma Cells-Cellular Accumulation and Cytotoxicity[Pubmed: 28665345] | | Abstract
Gliomas are one of the most aggressive and treatment-resistant types of human brain cancer. Identification and evaluation of anticancer properties of compounds found in plants, such as naringenin (N) and 8-Prenylnaringenin (8PN), are among the most promising applications in glioma therapy. The prenyl group seems to be crucial to the anticancer activity of flavones, since it may lead to enhanced cell membrane targeting and thus increased intracellular activity. It should be noted that 8PN content in hop cones is 10 to 100 times lower compared to other flavonoids, such as xanthohumol. In the study presented, we used a simple method for the synthesis of 8PN from isoxanthohumol-O-demethylation, with a high yield of 97%. Cellular accumulation and cytotoxicity of naringenin and 8-Prenylnaringenin in normal (BJ) and cancer cells (U-118 MG) was also examined. Obtained data indicated that 8-Prenylnaringenin exhibited higher cytotoxicity against used cell lines than naringenin, and the effect of both flavones was stronger in U-118 MG cells than in normal fibroblasts. The anticancer properties of 8PN correlated with its significantly greater (37%) accumulation in glioblastoma cells than in normal fibroblasts. Additionally, naringenin demonstrated higher selectivity for glioblastoma cells, as it was over six times more toxic for cancer than normal cells. Our results provide evidence that examined prenylated and non-prenylated flavanones have different biological activities against normal and cancer cell lines, and this property may be useful in designing new anticancer drugs for glioblastoma therapy.
Keywords: 8-Prenylnaringenin; cellular accumulation; confocal microscopy; cytotoxicity; glioblastoma; naringenin. |
|


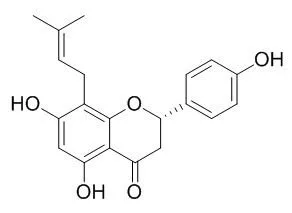



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)