| Description: |
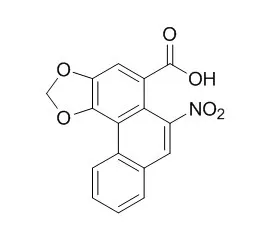
Aristolochic acid II (Aristolochic acid B,AAII), one of the major components of the carcinogenic plant extract aristolochic acid, is known to be mutagenic and to form DNA adducts in vitro and in vivo, AAII shows more carcinogenic risk than aristolochic acid I, and this may be, at least partly, the result of its increased levels in kidney and plasma. |
| In vitro: |
| Am J Chin Med. 2008;36(2):425-36. | | Production, characterization of a monoclonal antibody against aristolochic acid-B and development of its assay system.[Pubmed: 18457371] | Aristolochic acid-II (Aristolochic acid B, AA-II) conjugated with bovine serum albumin (BSA) was used as an antigen for immunizing BALB/c mice.
METHODS AND RESULTS:
Isolated splenocytes from the immunized mice were fused with an aminopterin-sensitive mouse myeloma cell line, SP2/0-Ag14, to produce hybridoma cells that secreted a monoclonal antibody (MAb) against AA-II. The selected hybridoma was subsequently cloned by limited dilution method. For MAb, the isotype and an estimated dissociation constant (K(D)) of the MAb were determined. The MAb was used to establish an ELISA method. Accuracy and variation assays, as well as determinations of the specificity and sensitivity, were also carried out and the linear range was 0.19-13 microg/ml. The anti-AA-II MAb showed a very high specificity for AA-II and had low cross-reactivities against the other aristolochic acid (AAs) (CR: AA-I, 3.4%; AA-VIIa, 0.86%) or aristololactam-I (AL-I) (CR<0.07%) except AA-IIIa which has 17% of cross activity. Anti-AA-II MAb also showed negligible cross-reactivity (<0.5%) toward other natural compounds with different chemical structures including barbaloin, sennoside A, rutin, glycyrrhizin, caffeic acid etc.
CONCLUSIONS:
This is the first time that an ELISA method was successfully established for the application of anti-AA-II MAb. |
|
| In vivo: |
| Mutat Res. 2012 Mar 18;743(1-2):52-8. | | Comparison of the mutagenicity of aristolochic acid I and aristolochic acid II in the gpt delta transgenic mouse kidney.[Pubmed: 22245565 ] | Aristolochic acid (AA) is known to be a potent mutagen and carcinogen. Aristolochic acid I (Aristolochic acid A, AAI) and aristolochic acid II (Aristolochic acid B, AAII), the two major components of AA, differ from each other by a single methoxy group. However, their individual mutagenic characteristics in vivo are unclear.
METHODS AND RESULTS:
In the present study, we compared their DNA adduct formation and mutagenicities in the gpt delta transgenic mouse kidney. The dA-AAI, dG-AAI, dA-AAII and dG-AAII were identified in the kidney two days after intragastric administration of AAI or AAII at 5mg/kg. The concentration of DNA adducts formed by AAII was approximately 2.5-fold higher than that formed by AAI (p<0.05). The mutant frequency induced by AAII was nearly two-fold higher than that induced by AAI (p<0.05) following administration of 5mg/kg AAI or AAII, five times per week for six weeks. Investigation of the mutation spectra showed no statistically significant difference between AAI- and AAII-treated mice (p>0.05). A:T to T:A transversion was the predominant type of mutation in both treated groups, the GC-associated mutation rates, however, differed between the AAI and AAII treatments. The in vivo metabolic pathways of AAI and AAII are different, and this may affect their mutagenicity. In the present study, we measured the levels of AAI and AAII in the kidney and plasma of gpt delta transgenic mice at multiple time points after a single intragastric dose of 1 or 5mg/kg of either component. Our results showed that the levels of AAII in both kidney and plasma were considerably higher than those of AAI (p<0.01).
CONCLUSIONS:
The present study indicated that AAII showed more carcinogenic risk than AAI in vivo, and this may be, at least partly, the result of its increased levels in kidney and plasma. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)