Natural Products

- ChemFaces is a professional high-purity natural products manufacturer.
- Product Intended Use
- 1. Reference standards
- 2. Pharmacological research
- 3. Inhibitors



| Size /Price /Stock | 10 mM * 1 mL in DMSO / Inquiry | Other Packaging | *Packaging according to customer requirements(100uL/well, 200uL/well and more), and Container use Storage Tube With Screw Cap |

| Size /Price /Stock | 10 mM * 100 uL in DMSO / Inquiry / In-stock 10 mM * 1 mL in DMSO / Inquiry / In-stock | Related Libraries |
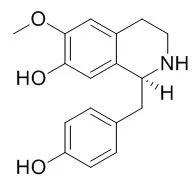
| Description: | (+)-R-Coclaurine and (+)-S-reticuline show negative inotropic effects, coclaurine derivatives and of paeoniflorin derivatives have neuromuscular blocking actions. D-Coclaurine has a neuroleptic-like property in blocking effects of dopaminergic stimulating agents. | ||||||
| Targets: | Calcium Channel | ||||||
| In vitro: |
| ||||||
| In vivo: |
|
| Source: | The roots of Coptis chinensis Franch |
| Solvent: | Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| Storage: | Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to 24 months(2-8C). Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour. Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com |
| After receiving: | The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling. |
 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.5051 mL | 17.5254 mL | 35.0508 mL | 70.1016 mL | 87.6271 mL |
| 5 mM | 0.701 mL | 3.5051 mL | 7.0102 mL | 14.0203 mL | 17.5254 mL |
| 10 mM | 0.3505 mL | 1.7525 mL | 3.5051 mL | 7.0102 mL | 8.7627 mL |
| 50 mM | 0.0701 mL | 0.3505 mL | 0.701 mL | 1.402 mL | 1.7525 mL |
| 100 mM | 0.0351 mL | 0.1753 mL | 0.3505 mL | 0.701 mL | 0.8763 mL |
| Structure Identification: |
|