| Description: |
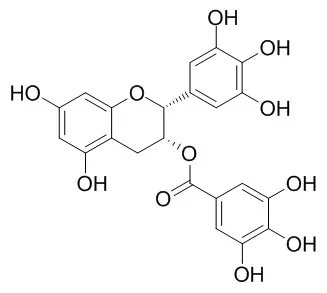
(-)-Epigallocatechin Gallate is an antioxidant polyphenol flavonoid form green tea, and inhibits the activation of EGFR, HER2 and HER3, with antitumor activity. (-)-Epigallocatechin gallate treatment enduring to cardio protection at mitochondrial level, it has protective effects against βA-induced neuronal apoptosis through scavenging reactive oxygen species, which may be beneficial for the prevention of Alzheimer's disease. |
| Targets: |
Caspase | Bcl-2/Bax | Beta Amyloid | p53 | COX | Bcl-XL |
| In vitro: |
| Mol Biol Rep. 2013 Dec;40(12):6533-45. | | (-)-Epigallocatechin-gallate (EGCG) stabilize the mitochondrial enzymes and inhibits the apoptosis in cigarette smoke-induced myocardial dysfunction in rats.[Pubmed: 24197690] |
METHODS AND RESULTS:
The present study brings out the preventive role of (-)-Epigallocatechin gallate (EGCG) on cardiac mitochondrial metabolism and apoptosis in cigarette smoke (CS)-exposed rats. The CS-exposed rats showed significantly decreased activities of TCA cycle enzymes and mitochondrial enzymatic antioxidants, on the other hand, mitochondrial lipid peroxidation was increased and GSH level was decreased. Further, CS exposure was found to induce cardiac apoptosis through release of cytochrome c into the cytosol, cleavage of pro-caspase-3 to active caspase-3, up-regulation of pro-apoptotic (Bax) and down-regulation of antiapoptotic (Bcl-2) molecules. The CS-induced apoptosis was further confirmed by mitochondrial and nuclear ultra structural apoptotic features as evaluated by electron microscopic studies. (-)-Epigallocatechin gallate supplementation shelters the activities of TCA cycle enzymes and antioxidant enzymes, with concomitant decrease in lipid peroxidation and increase in GSH level. (-)-Epigallocatechin gallate administration inhibited apoptosis through the inhibition of cytochrome c release into cytosol, activation of pro-caspase-3, down regulation of Bax and significant up regulation of Bcl-2. (-)-Epigallocatechin gallate reversed the ultra structural apoptotic alterations of mitochondria and nucleus.
CONCLUSIONS:
The present study has provided experimental evidences that the (-)-Epigallocatechin gallate treatment enduring to cardio protection at mitochondrial level. |
|
| In vivo: |
| Endocrinology, 2000, 141(3):980-7. | | Modulation of endocrine systems and food intake by green tea epigallocatechin gallate.[Pubmed: 10698173] | Green tea polyphenols, especially the catechin, (-)-Epigallocatechin gallate (EGCG), have been proposed as a cancer chemopreventative based on a variety of laboratory studies.
METHODS AND RESULTS:
For clear assessment of the possible physiological effects of green tea consumption, we injected pure green tea catechins ip into rats and studied their acute effects on endocrine systems. We found that EGCG, but not related catechins, significantly reduced food intake; body weight; blood levels of testosterone, estradiol, leptin, insulin, insulin-like growth factor I, LH, glucose, cholesterol, and triglyceride; as well as growth of the prostate, uterus, and ovary. Similar effects were observed in lean and obese male Zucker rats, suggesting that the effect of EGCG was independent of an intact leptin receptor. EGCG may interact specifically with a component of a leptin-independent appetite control pathway.
Endocrine changes induced by parenteral administration of EGCG may relate to the observed growth inhibition and regression of human prostate and breast tumors in athymic mice treated with EGCG as well as play a role in the mechanism by which EGCG inhibits cancer initiation and promotion in various animal models of cancer. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)