| In vitro: |
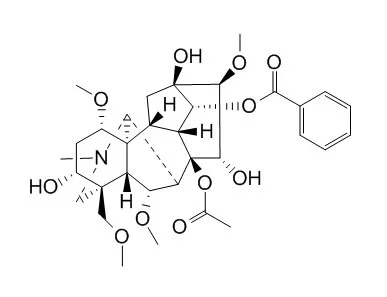
| Eur J Pharmacol. 2002 Feb 2;436(3):217-25. | | Mesaconitine-induced relaxation in rat aorta: involvement of Ca2+ influx and nitric-oxide synthase in the endothelium.[Pubmed: 11858801] | Aconiti tuber, roots of aconite (Aconitum japonicum), is an oriental herbal medicine used for centuries in Japan and China to improve the health of persons with a weak constitution and poor metabolism.
METHODS AND RESULTS:
We investigated the effects of Mesaconitine, one of the aconite alkaloids in Aconiti tuber, on the contraction and free intracellular Ca2+ concentration ([Ca2+]i) level in isolated rat thoracic aorta. Mesaconitine at 30 microM inhibited 3 microM phenylephrine-induced contraction in the endothelium-intact, but not endothelium-denuded, aortic rings. The effect of Mesaconitine was dependent on external Ca2+ concentrations. The relaxation induced by Mesaconitine was abolished by N(omega)-nitro-L-arginine methyl ester (0.1 mM, an inhibitor of nitric-oxide synthase), as well as the relaxation induced by acetylcholine. Acetylcholine induced relaxation in two phases in our conditions; the initial phase was transient and external Ca2+ -independent, and the second phase was sustained and external Ca2+ -dependent. Treatment with 100 nM thapsigargin, which depleted intracellular Ca2+ stores, inhibited acetylcholine-induced, but not Mesaconitine-induced, relaxation. Mesaconitine increased the [Ca2+]i level in endothelial cells by influx of Ca2+ from extracellular spaces.
CONCLUSIONS:
These findings suggest that Mesaconitine-induced Ca2+ influx and activation of nitric-oxide synthase in endothelial cells and, thus, induced vasorelaxation in rat aorta. | | Eur J Pharmacol. 1998 Jan 26;342(2-3):183-91. | | Inhibition of stimulus-triggered and spontaneous epileptiform activity in rat hippocampal slices by the Aconitum alkaloid mesaconitine.[Pubmed: 9548384] | The aim of the present study was to investigate if the plant alkaloid, Mesaconitine, which has been reported to have antinociceptive effects via stimulation of the noradrenergic system, inhibits epileptiform field potentials.
METHODS AND RESULTS:
The experiments were performed as extracellular recordings on rat hippocampal slices. Epileptiform activity was induced by omission of Mg2+ from the bathing medium or by addition of bicuculline and stimulus-evoked population bursts were recorded in the CA1 region. Spontaneous epileptiform activity was elicited by perfusing a nominally Mg2+-free bathing medium with high K+ concentration (5 mM). Both stimulus-triggered and spontaneous epileptiform activity was attenuated in a concentration-dependent manner by Mesaconitine (30 nM-1 microM). The inhibitory effect was rather variable in appearance when lower concentrations (30 and 100 nM) of Mesaconitine were applied. Pretreatment of the slices with the alpha-adrenoceptor antagonist yohimbine (1 microM) prevented the effect of Mesaconitine.
CONCLUSIONS:
It is concluded that the inhibitory action of Mesaconitine at low concentration is mediated via alpha-adrenoceptors. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)