| Structure Identification: |
| Pharm Biol. 2013 Sep;51(9):1204-7. | | Cytotoxic and potential anticancer constituents from the stems of Schisandra pubescens.[Pubmed: 23883077 ] | The diethyl ether extract of the stems of Schisandra pubescens Hemsl. et Wils. (Schisandraceae) was found to exhibit cytotoxic activity in vitro. However, investigations of the bioactive constituents of this plant have been very limited.
Elucidation of the cytotoxic constituents of S. pubescens was performed.
METHODS AND RESULTS:
Repeated silica gel column chromatography and preparative TLC were used for the chemical investigation of the diethyl ether extract of S. pubescens stems. All isolates were evaluated for their in vitro cytotoxicity against A549, PC-3, KB and KBvin human cancer cell lines.
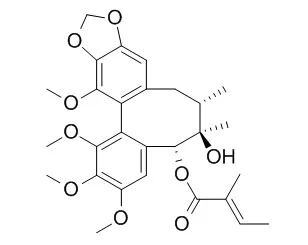
Nine known compounds were obtained, including four lignans, epischisandrone (1), Tigloylgomisin P (2), cagayanone (3) and (-)-gomisin L₂ (4), together with five triterpenoids, micranoic acid B (5), lancifodilactone H (6), coccinic acid (7), schisanlactone B (8) and anwuweizonic acid (9). Compounds 2-6 and 8 showed moderate to marginal cytotoxicity, with GI₅₀ values of 11.83-35.65 μM.
CONCLUSIONS:
The isolation of 1-9 from S. pubescens and the cytotoxicities of 3-6 are first reported. Compounds 2-6 and 8 could be the active principles responsible for the anticancer effects of S. pubescens. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)