| In vitro: |
| Molecules. 2013 Feb 4;18(2):1985-94. | | Cytotoxicity and structure-activity relationships of xanthone derivatives from Mesua beccariana, Mesua ferrea and Mesua congestiflora towards nine human cancer cell lines.[Pubmed: 23381024] |
The cytotoxic structure-activity relationships among a series of xanthone derivatives from Mesua beccariana, Mesua ferrea and Mesua congestiflora were studied.
METHODS AND RESULTS:
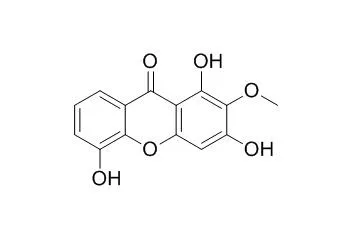
Eleven xanthone derivatives identified as mesuarianone (1), mesuasinone (2), mesuaferrin A (3), mesuaferrin B (4), mesuaferrin C (5), 6-deoxyjacareubin (6), caloxanthone C (7), macluraxanthone (8), 1,5-dihydroxyxanthone (9), Tovopyrifolin C (10) and α-mangostin (11) were isolated from the three Mesua species. The human cancer cell lines tested were Raji, SNU-1, K562, LS-174T, SK-MEL-28, IMR-32, HeLa, Hep G2 and NCI-H23. Mesuaferrin A (3), macluraxanthone (8) and α-mangostin (11) showed strong cytotoxicities as they possess significant inhibitory effects against all the cell lines.
CONCLUSIONS:
The structure-activity relationship (SAR) study revealed that the diprenyl, dipyrano and prenylated pyrano substituent groups of the xanthone derivatives contributed towards the cytotoxicities. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)