| Structure Identification: |
| Helvetica Chimica Acta, 2013 , 96 (2) :313-9. | | Two New Abietane Diterpenoids from the Roots of Tripterygium wilfordii HOOK. f.[Reference: WebLink] |
METHODS AND RESULTS:
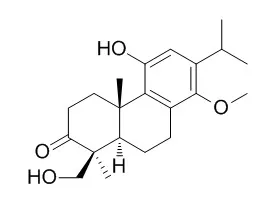
Two new abietane-type diterpenoids, named triptobenzene R (1) and triptobenzene S (2), together with three known abietane-type diterpenoids, triptophenolide (3), Triptonodiol (4), and triptonoterpene methyl ether (5), were isolated from the roots of Tripterygium wilfordii Hook. f. Their structures and relative configurations were established by detailed spectral studies, including 1D- and 2D-NMR (HSQC, HMBC, and NOESY), and HR-ESI-TOF-MS, and by comparison with published data.
CONCLUSIONS:
Their absolute configurations were assigned by the CD technique, applied for the first time to abietane diterpenes from Tripterygium wilfordii. Compound 2 is the first abietane-type norditerpenoid isolated from the genus Tripterygium. | | Planta Med. 1988 Aug;54(4):330-2. | | Studies on New Components and Stereochemistry of Diterpenoids from Trypterygium wilfordii.[Pubmed: 17265278 ] | | An extensive investigation of the petroleum ether extract of TRIPTERYGIUM WILFORDII plants have revealed the presence of four new compounds, triptonoterpene, neotriptonoterpene, Triptonodiol, and neotriptonolide, in addition to the diterpenes isolated and characterized earlier. The structures of these novel natural products have been elucidated. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)