METHODS AND RESULTS:
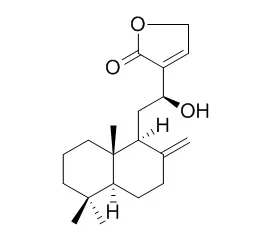
Bioassay-guided purification of the dichloromethane extract of the leaves of the Malaysian species Vitex vestita, led to the isolation of six new labdane-type diterpenoids, namely, 12-epivitexolide A (2), vitexolides B and C (3 and 4), vitexolide E (8), and vitexolins A and B (5 and 6), along with six known compounds, vitexolide A (1) and Vitexolide D (7), acuminolide (9), 3β-hydroxyanticopalic acid (10), 8α-hydroxyanticopalic acid (11), and 6α-hydroxyanticopalic acid (12). Their structures were elucidated on the basis of 1D and 2D NMR analyses and HRMS experiments. Both variable-temperature NMR spectroscopic studies and chemical modifications were performed to investigate the dynamic epimerization of the γ-hydroxybutenolide moiety of compounds 1-4. Compounds were assayed against a panel of 46 Gram-positive strains.
CONCLUSIONS:
Vitexolide A (1) exhibited the most potent antibacterial activity with minimal inhibitory concentration values ranging from 6 to 96 μM, whereas compounds 2 and 6-9 showed moderate antibacterial activity. The presence of a β-hydroxyalkyl-γ-hydroxybutenolide subunit contributed significantly to antibacterial activity. Compounds 1-4 and 6-9 showed cytotoxic activities against the HCT-116 cancer cell line (1 < IC50s < 10 μM) and human fetal lung fibroblast MRC5 cell line (1 < IC50s < 10 μM for compounds 1, 2, 7, 8, and 9). |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)