- ChemFaces is a professional high-purity natural products manufacturer.
- Product Intended Use
- 1. Reference standards
- 2. Pharmacological research
- 3. Inhibitors
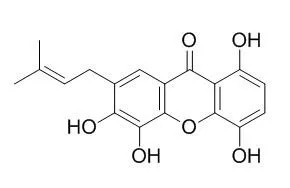
1,4,5,6-Tetrahydroxy-7-prenylxanthone
ChemFaces products have been cited in many studies from excellent and top scientific journals
Contact Us
Order & Inquiry & Tech Support
Tel: (0086)-27-84237683
Tech: service@chemfaces.com
Order: manager@chemfaces.com
Address: 176, CheCheng Eest Rd., WETDZ, Wuhan, Hubei 430056, PRC
How to Order
Orders via your E-mail:
1. Product number / Name / CAS No.
2. Delivery address
3. Ordering/billing address
4. Contact information
Order: manager@chemfaces.com
Delivery time
Delivery & Payment method
1. Usually delivery time: Next day delivery by 9:00 a.m. Order now
2. We accept: Wire transfer & Credit card & Paypal
Citing Use of our Products
* Packaging according to customer requirements(5mg, 10mg, 20mg and more). We shipped via FedEx, DHL, UPS, EMS and others courier.
According to end customer requirements, ChemFaces provide solvent format. This solvent format of product intended use: Signaling Inhibitors, Biological activities or Pharmacological activities.
| Size /Price /Stock |
10 mM * 1 mL in DMSO / Inquiry |
Other Packaging |
*Packaging according to customer requirements(100uL/well, 200uL/well and more), and Container use Storage Tube With Screw Cap |
More articles cited ChemFaces products.
- Cell Physiol Biochem....2017...
- BMC Complement Med Ther. ...2020...
- Reprod Toxicol.2020, 96:1-10.
- Nutrients.2019, 12(1):E40
- J Ethnopharmacol.2019, 241:112025
- J Agric Food Chem.2015, 63(44):9869-78
- Cell Rep.2022, 39(1):110643.
- J Biomol Struct Dyn.2022, 5;1-17.
- Molecules.2020, 25(20):4851.
- Antioxidants (Basel).2021, 10(3):379.
- Bull. Natl. Mus. Nat. Sci....2021...
- Molecules.2022, 27(22):7887.
- Antioxidants (Basel).2021, 10(10):1620.
- Polytechnic University of Catalon...2017...
- Biochem Systematics and Ecology...2017...
- Plant Pathology2022, 13527
- Institut Pasteur Korea...2020...
- Nutr Metab (Lond).2019, 16:31
- Heliyon2022, 8(2):e08866.
- Int J Mol Sci.2021, 22(14):7324.
- J of Essential Oil Research...2019...
- Phytomedicine2022, 104:154337.
- Phys Chem Chem Phys....2018...
- More...
Our products had been exported to the following research institutions and universities, And still growing.
- Universidad de Buenos Aires (Argentina)
- University of Vienna (Austria)
- University of Madras (India)
- National Hellenic Research Foun... (Greece)
- Srinakharinwirot University (Thailand)
- Technical University of Denmark (Denmark)
- Mahidol University (Thailand)
- Instituto de Investigaciones Ag... (Chile)
- FORTH-IMBB (Greece)
- Instytut Nawozów Sztucznych w ... (Poland)
- Sanford Burnham Medical Researc... (USA)
- More...
- In Vitro Cellular & Developmental Biology - Plant2022, 58:972-988.
- J Ethnopharmacol.2017, 209:305-316
- Eur J Pharmacol.2021, 899:174010.
- Vietnam Journal of Food Control.2022, 5(2): 115-128.
- Plants2022, 11(3),294.
- Molecules.2020, 25(15):3353.
- J Pharmaceut Biomed2020, 182:113110
- Inflammation.2021, doi: 10.1007
- Front Pharmacol.2020, 11:251.
- Biomedicine & Pharmacotherapy2022, 153:113404.
Related Screening Libraries
| Size /Price /Stock |
10 mM * 100 uL in DMSO / Inquiry / In-stock
10 mM * 1 mL in DMSO / Inquiry / In-stock
|
Related Libraries |
|
| Description: |
1,4,5,6-Tetrahydroxy-7-prenylxanthone has anti-cancer activity, it shows moderate cytotoxicities against breast cancer (MDA-MB-435S) and lung adenocarcinoma (A549) cell lines; it also exhibits moderate activities with GI50 (Growth inhibitory) values of 2.8 μM against the human leukaemic HL-60 cell line were measured in vitro. |
| In vitro: |
| Phytochemistry. 2009 Jan;70(1):60-8. | | Xanthone biosynthesis in Hypericum perforatum cells provides antioxidant and antimicrobial protection upon biotic stress.[Pubmed: 19062051 ] |
METHODS AND RESULTS:
Methanolic extract of elicited cells exhibited significantly higher antioxidant and antimicrobial competence than the equivalent extract of control HP cells indicating that these properties have been significantly increased in HP cells after elicitation. Four major de novo synthesized xanthones have been identified as 1,3,6,7-tetrahydroxy-8-prenyl xanthone(1,4,5,6-Tetrahydroxy-7-prenylxanthone), 1,3,6,7-tetrahydroxy-2-prenyl xanthone, 1,3,7-trihydroxy-6-methoxy-8-prenyl xanthone and paxanthone.
CONCLUSIONS:
Antioxidant and antimicrobial characterization of these de novo xanthones have revealed that xanthones play dual function in plant cells during biotic stress: (1) as antioxidants to protect the cells from oxidative damage and (2) as phytoalexins to impair the pathogen growth. |
|
1,4,5,6-Tetrahydroxy-7-prenylxanthone Description
| Source: |
The twig barks of Garcinia xanthochymus |
| Solvent: |
Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| Storage: |
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to 24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
|
| After receiving: |
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling. |
ChemFaces New Products and Compounds
Recently, ChemFaces products have been cited in many studies from excellent and top scientific journals

Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.
IF=36.216(2019)PMID: 29328914
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.
IF=22.415(2019)PMID: 32004475
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.
IF=14.548(2019)PMID: 29149595
ACS Nano. 2018 Apr 24;12(4): 3385-3396. doi: 10.1021/acsnano.7b08969.
IF=13.903(2019)PMID: 29553709
Nature Plants. 2016 Dec 22;3: 16206. doi: 10.1038/nplants.2016.205.
IF=13.297(2019)PMID: 28005066
Sci Adv. 2018 Oct 24;4(10): eaat6994. doi: 10.1126/sciadv.aat6994.
IF=12.804(2019)PMID: 30417089
Calculate Dilution Ratios(Only for Reference)
|
1 mg |
5 mg |
10 mg |
20 mg |
25 mg |
| 1 mM |
3.046 mL |
15.23 mL |
30.4599 mL |
60.9199 mL |
76.1499 mL |
| 5 mM |
0.6092 mL |
3.046 mL |
6.092 mL |
12.184 mL |
15.23 mL |
| 10 mM |
0.3046 mL |
1.523 mL |
3.046 mL |
6.092 mL |
7.615 mL |
| 50 mM |
0.0609 mL |
0.3046 mL |
0.6092 mL |
1.2184 mL |
1.523 mL |
| 100 mM |
0.0305 mL |
0.1523 mL |
0.3046 mL |
0.6092 mL |
0.7615 mL |
* Note: If you are in the process of experiment, it's need to make the dilution ratios of the samples. The dilution data of the sheet for your reference. Normally, it's can get a better solubility within lower of Concentrations.
Tags: buy 1,4,5,6-Tetrahydroxy-7-prenylxanthone | 1,4,5,6-Tetrahydroxy-7-prenylxanthone supplier | purchase 1,4,5,6-Tetrahydroxy-7-prenylxanthone | 1,4,5,6-Tetrahydroxy-7-prenylxanthone cost | 1,4,5,6-Tetrahydroxy-7-prenylxanthone manufacturer | order 1,4,5,6-Tetrahydroxy-7-prenylxanthone | 1,4,5,6-Tetrahydroxy-7-prenylxanthone distributor






 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)