| Structure Identification: |
| J Agric Food Chem. 2009 Aug 26;57(16):7480-9. | | Identification and RP-HPLC-ESI-MS/MS quantitation of bitter-tasting beta-acid transformation products in beer.[Pubmed: 19627140 ] |
METHODS AND RESULTS:
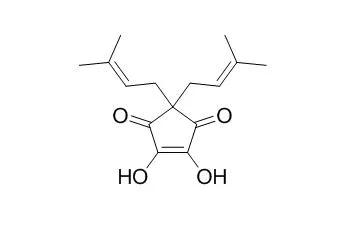
Thermal treatment of the hop beta-acid colupulone under wort boiling conditions, followed by LC-TOF-MS and 1D/2D NMR spectroscopy, revealed cohulupone, Hulupinic acid, nortricyclocolupone, two tricyclocolupone epimers, two dehydrotricyclocolupone epimers, two hydroxytricyclocolupone epimers, and two hydroperoxytricyclocolupone epimers as the major bitter-tasting beta-acid transformation products. Among these compounds, the chemical structures of the hydroxy- as well as the hydroperoxytricyclocolupone epimers have not previously been confirmed by 1D/2D NMR experiments. Depending on their chemical structure, these compounds showed rather low recognition thresholds ranging from 7.9 to 90.3 micromol/L.
The lowest thresholds of 7.9 and 14.7 micromol/L were found for cohulupone, imparting a short-lasting, iso-alpha-acid-like bitter impression, and for hydroxytricyclocolupone, exhibiting a long-lasting, lingering, and harsh bitterness perceived on the posterior tongue and throat. Furthermore, HPLC-ESI-MS/MS analysis allowed for the first time a simultaneous detection and quantitation of these bitter-tasting beta-acid transformation products in a range of commercial beer samples without any sample cleanup.
CONCLUSIONS:
Depending on the type of beer, these studies revealed remarkable differences in the concentrations of the individual beta-acid transformation products. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)