| Arch Pharm Res. 2005 Sep;28(9):1031-6. |
| Antimicrobial property of (+)-lyoniresinol-3alpha-O-beta-D-glucopyranoside isolated from the root bark of Lycium chinense Miller against human pathogenic microorganisms.[Pubmed: 16212233] |
METHODS AND RESULTS:
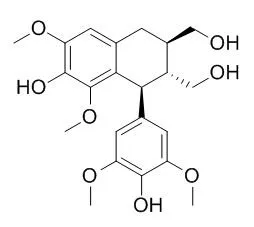
(+)-Lyoniresinol-3alpha-O-beta-D-glucopyranoside (1) was isolated from an ethyl acetate extract of the root bark from Lycium chinense Miller, and its structure was determined using 1D and 2D NMR spectroscopy including DEPT, HMQC, and HMBC. (+)-Lyoniresinol-3alpha-O-beta-D-glucopyranoside exhibited potent antimicrobial activity against antibiotic-resistant bacterial strains, methicillin-resistant Staphylococcus aureus (MRSA) isolated from patients, and human pathogenic fungi without having any hemolytic effect on human erythrocytes. In particular, compound 1 induced the accumulation of intracellular trehalose on C. albicans as stress response to the drug, and disrupted the dimorphic transition that forms pseudo-hyphae caused by the pathogenesis.
CONCLUSIONS:
This indicates that (+)-Lyoniresinol-3alpha-O-beta-D-glucopyranoside has excellent potential as a lead compound for the development of antibiotic agents. |
| Mol Cell Biochem. 2013 Jan;373(1-2):211-6. |
| Lyoniresinol inhibits melanogenic activity through the induction of microphthalmia-associated transcription factor and extracellular receptor kinase activation.[Pubmed: 23099844 ] |
Lyoniresinol was one of the eight lignans purified from methanol extract of Vitex negundo and was shown to have robust tyrosinase inhibitory capacity. However, the mechanism of its action is not known.
METHODS AND RESULTS:
Hence, the goal of the current study was to study the effects of Lyoniresinol on cytotoxicity and melanin content in murine B16F10 melanoma cells and to delineate the underlying mechanism of tyrosinase inhibition. Lyoniresinol was purified from methanol extract of Vitex negundo root and when tested in B16F10, cells showed robust anti-melanogenic activity. It caused the downregulation of microphthalmia-associated transcription factor (MITF) and tyrosinase steady state protein expression levels. Flow cytometry analysis of Lyoniresinol-treated cells showed that the latter activates extracellular receptor kinase (ERK) phosphorylation, which causes MITF protein degradation and suppression of tyrosinase activity. Lyoniresinol decreased tyrosinase activity and melanin biosynthesis in B16F10 cells by activating ERK signaling, which downregulated MITF, tyrosinase, but not TRP-1 and TRP-2 protein expression.
CONCLUSIONS:
Contingent to more vigorous in vitro and in vivo experiments, Lyoniresinol can perhaps be incorporated into clinical dermatologic use as a skin lightening agent. |
| West African J. Med., 1994, 13(1):495-8. |
| Changes in Lyoniresinol Content and Antioxidative Activity of Pickled Ume during the Seasoning Process[Reference: WebLink] |
Lyoniresinol content and antioxidative activity of commercially available seasoning pickled Ume was investigated.
METHODS AND RESULTS:
The seasoning pickled Ume with low salt concentration showed relatively low lyoniresinol content and antioxidative activity compared to products with high salt concentrations. This result indicates that excessive de-salt operation leads to decrease of a lyoniresinol content and antioxidative activity of flesh. Although remarkable decrease of lyoniresinol content and antioxidative activity of pickled Ume was observed in the early stage of seasoning process, re-extraction of lyoniresinol and antioxidative activity by osmotic pressure of seasoning liquid was thought to occur after two weeks and later.
CONCLUSIONS:
From the results obtained, we concluded that enough period seasoning by the seasoning liquid with enough osmotic pressure is necessary to produce the seasoning pickled Ume with rich nutritional function. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)