| In vitro: |
| J Ethnopharmacol. 2015 Nov 4;174:57-65. | | In vitro opioid receptor affinity and in vivo behavioral studies of Nelumbo nucifera flower.[Pubmed: 26260436 ] | Nelumbo nucifera Geartn., known as sacred lotus, has been used traditionally in South East Asia as a traditional medicine for various CNS disorders including stress, fever, depression, insomnia, and cognitive conditions.
To investigate the in vitro cannabinoid and opioid receptor binding affinities, and in vivo behavioral actions of Nelumbo flower extracts and to isolate the potential compounds to treat CNS associated disorders.
METHODS AND RESULTS:
The white and pink flowers of N. nucifera were extracted with 95% EtOH, followed by acid-base partitioning using CHCl3 to give acidic and basic partitions. These partitions were subjected to Centrifugal Preparative TLC (CPTLC) to yield benzyltetrahydroisoquinoline (BTIQ) alkaloids and long chain fatty acids, identified by physical and spectroscopic methods. In addition, EtOH extracts and partitions were analyzed for chemical markers by UHPLC/MS and GC/MS. In vitro neuropharmacological effects were evaluated by cannabinoid (CB1 and CB2) and opioid [delta (δ), kappa (ĸ), and mu (µ)] competitive radioligand binding and GTPγS functional assays. The in vivo behavioral effect was studied through the use of the mouse tetrad assay at 10, 30, 75 and 100mg/kg/ip doses that revealed the effect on locomotion, catalepsy, body temperature, and nociception of acidic and basic CHCl3 partitions, fractions, and compounds.
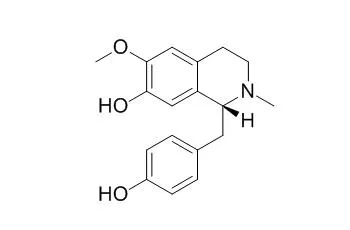
Three aporphines, nuciferine (1), N-nor-nuciferine (2), asimilobine (3), and five BTIQs, armepavine (4), O-methylcoclaurine (5), N-Methylcoclaurine (6), coclaurine (7), neferine (10), and a mixture of linoleic and palmitic acids (LA and PA), were identified and evaluated for cannabinoid and opioid receptor displacement activities. Compounds 5-7 showed binding affinities for the ĸ opioid receptor with equilibrium dissociation constant (Ki) values of 3.5 ± 0.3, 0.9 ± 0.1, 2.2 ± 0.2 μM, respectively. Compound 10 displayed affinities for δ-and µ- opioid receptors with Ki values of 0.7 ± 0.1 and 1.8 ± 0.2 μM, respectively, and was determined to be a weak δ agonist by GTPγS functional assay. The mixture of LA and PA (1:1) showed an affinity for δ opioid receptor with a Ki value of 9.2 ± 1.1 μM. The acidic and basic CHCl3 partitions, compounds 1 and 7, and 5-7 mixture were subjected to the tetrad assay, of which the acidic partition displayed decreased locomotion and increased catalepsy, antinociception, and hypothermia in animal at doses of 75-100 mg/kg/ip, and also showed clonic-tonic seizures upon touch at 100mg/kg.
CONCLUSIONS:
Bioassay-guided isolation revealed compounds 5-7, 10, and the mixture of LA and PA displayed various degrees of opioid receptor radioligand displacement affinities. The in vivo tetrad assay of acidic CHCl3 partition, enriched with aporphines 1 and 2, displayed actions on all four points of behavioral parameters. It can be concluded that the in vivo mild canabimimetic-type effect observed for the CHCl3 partition is likely mediated through other CNS mechanisms since the extracts, partitions, and isolated compounds had no affinity for the in vitro CB1 and CB2 receptors. This work, along with traditional use and the reported bioactivities of the BTIQ alkaloids, suggested further studies on N. nucifera are needed to understand the roles that the extracts and/or individual compounds might contribute to the behavioral effects. | | Nat Prod Commun. 2015 Apr;10(4):577-80. | | Alkaloids from Peumus boldus and their acetylcholinesterase, butyrylcholinesterase and prolyl oligopeptidase inhibition activity.[Pubmed: 25973480] | Eleven isoquinoline alkaloids (1-11) were isolated from dried leaves of Peumus boldus Mol. by standard chromatographic methods.
METHODS AND RESULTS:
The chemical structures were elucidated by MS, and 1D and 2D NMR spectroscopic analysis, and by comparison with literature data. Compounds isolated in sufficient amount were evaluated for their acetylcholinesterase, and butyrylcholinesterase inhibition activity using Ellman's method. In the prolyl oligopeptidase assay, Z-Gly-Pro-p-nitroanilide was used as substrate. Promising butyrylcholinesterase inhibition activities were demonstrated by two benzylisoquinoline alkaloids, reticuline (8) and N-Methylcoclaurine (9), with IC50 values of 33.6 ± 3.0 μM and 15.0 ± 1.4 μM, respectively.
CONCLUSIONS:
Important prolyl oligopeptidase inhibition activities were shown by N-methyllaurotetanine (6) and sinoacutine (4) with IC50 values of 135.4 ± 23.2 μM and 143.1 ± 25.4 μM, respectively. Other tested compounds were considered inactive. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)