| Structure Identification: |
| Chem Pharm Bull (Tokyo). 1993 Mar;41(3):566-70. | | Comparative studies on the constituents of ophiopogonis tuber and its congeners. VIII. Studies on the glycosides of the subterranean part of Ophiopogon japonicus Ker-Gawler cv. Nanus.[Pubmed: 8477511] |
METHODS AND RESULTS:
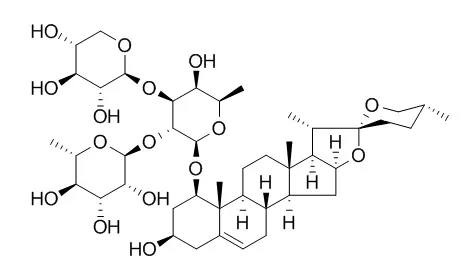
Two monoterpene glycosides, tentatively named OJV-I (1) and OJV-II (2), and eight steroidal glycosides, tentatively named OJV-III (3), OJV-IV (4), OJV-V (5), OJV-VI (6), OJV-VII (7), OJV-VIII (8), OJV-IX (9) and OJV-X (10), were isolated from the butanol-soluble fraction of the fresh subterranean part of Ophiopogon japonicus KER-GAWLER cv. Nanus.
CONCLUSIONS:
Among these compounds, 1, 2, 3, 4, 5, 6 and 7 were identified as l-borneo1 O-beta-D-glucopyranoside, l-borneo1 O-beta-D-apiofuranosyl (1-->6)-beta-D-glucopyranoside, ophiopogonin B, glycoside C, ophiopogonin D, Ls-10, and ruscogenin 1-O-sulfate, respectively.
The structures of compounds 8, 9, and 10 were established to be (23S,24S,25S)-23,24-dihydroxyruscogenin 1-O-[ alpha-L-rhamnopyranosyl(1-->2)] [beta-D-xylopyranosyl(1-->3)]-alpha-L-arabinopyranoside 24-O-beta-D-fucopyranoside, (23S,24S,25S)-23,24-dihydroxyruscogenin I-O-[alpha-L-2,3,4-tri-O-acetylrhamnopyranosyl(1-->2)][beta-D-xylo pyranosyl(1-->3)]-alpha-L-arabinopyranoside 24-O-beta-D-fucopyranoside, and (23S,24S,25S)-23,24-dihydroxyruscogenin 1-O-[alpha-L-2,3,4-tri-O-acetylrhamnopyranosyl(1-->2)] [beta-D-xylopyranosyl(1-->3)]=alpha-L-arabinopyranoside 24-O-beta-D-fucopyranoside, respectively. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)