| In vitro: |
| J. Asian Nat. Prod. Res.,2013;15(10):1130-8. | | Limonoid constituents of Euodia rutaecarpa var. bodinieri and their inhibition on NO production in lipopolysaccharide-activated RAW264.7 macrophages.[Pubmed: 23869424 ] |
METHODS AND RESULTS:
A new limonoid compound, named evorubodinin (1), was isolated from the dried and nearly ripe fruits of Euodia rutaecarpa (Juss.) Benth. var. bodinieri (Dode) Huang (family Rutaceae), together with two known limonoid compounds, limonin (2) and evolimorutanin (3). The chemical structure of 1 was elucidated by spectroscopic method and single-crystal X-ray diffraction. The inhibitory effects of the isolated compounds 1-3 and the structurally related compounds evodol (4), shihulimonin A1 (5), evodirutaenin (6), 12α-hydroxyRutaevin (7), and Rutaevin (8) on nitric oxide (NO) production in lipopolysaccharide-activated RAW264.7 macrophages were also assayed.
CONCLUSIONS:
All compounds 1-8 showed the inhibitory activity, in which both 7 and 8 with the uncommon 5β-H configuration more efficiently inhibited NO production. The results provided valuable information for further investigation of compounds 1-8 as anti-inflammatory agents or lead compounds. | | Chem. Biodivers.,2017 Mar;14(3) | | Pyranochromones from Dictyoloma vandellianum A. Juss and Their Cytotoxic Evaluation.[Pubmed: 27797447 ] |
METHODS AND RESULTS:
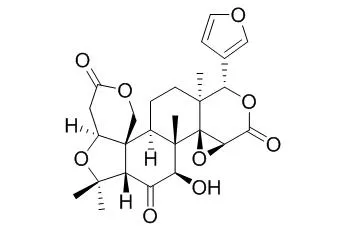
One new chromone 3,3-dimethylallylspatheliachromene methyl ether (1), as well as five known chromones, 6-(3-methylbut-2-enyl) allopteroxylin methyl ether (2), 6-(3-methylbut-2-enyl) allopteroxylin (3), 3,3-dimethylallylspatheliachromene (4), 5-O-methylcneorumchromone K (5) and spatheliabischromene (6), two alkaloids, 8-methoxy-N-methylflindersine (7) and 8-methoxyflindersine (8), and two limonoids, limonin diosphenol (9) and Rutaevin (10), were isolated from Dictyoloma vandellianum A. Juss (Rutaceae). Cytotoxic activities towards tumor cell lines B16-F10, HepG2, K562 and HL60 and non-tumor cells PBMC were evaluated for compounds 1 - 6.
CONCLUSIONS:
Compound 1 was the most active showing IC50 values ranging from 6.26 to 14.82 μg/ml in B16-F10 and K562 cell lines, respectively, and presented IC50 value of 11.65 μg/ml in PBMC cell line. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)