| Structure Identification: |
| Geochimica Et Cosmochimica Acta, 1989, 53(11):3073-3079. | | Tetrahymanol, the most likely precursor of gammacerane, occurs ubiquitously in marine sediments[Reference: WebLink] |
METHODS AND RESULTS:
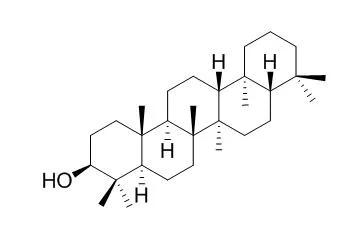
Tetrahymanol has been identified in several sediment samples from different depositional environments by gas chromatography-mass spectrometry and by coinjections with an authentic standard. Together with literature data this shows that Tetrahymanol is likely to be widespread, which is in accordance with the ubiquitous occurrence of its presumed diagenetic product, gammacerane, in more mature sediments and crude oils. The diagenetic conversion of Tetrahymanol to gammacerane most likely proceeds via dehydration and subsequent hydrogenation. The intermediate in this conversion, gammacer-2-ene, has been synthesized, and its presence in one sample confirmed by coinjections.
CONCLUSIONS:
The identification of Tetrahymanol in marine sediments indicates either that protozoa of the genus Tetrahymena are widely distributed or that Tetrahymanol is also a natural product of organisms other than Tetrahymena. | | Microbiology, 1990, 136(12):2551-2553. | | Tetrahymanol from the phototrophic bacterium Rhodopseudomonas palustris: First report of a gammacerane triterpene from a prokaryote[Reference: WebLink] | | Tetrahymanol, a rare triterpene from the gammacerane series, previously detected only in a few scattered eukaryotic taxa, has been isolated for the first time from the phototrophic bacterium Rhodopseudomonas palustris. This extends the significance of geochemical markers from this series, which are abundant in marine sediments and palaeohypersaline environments. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)