| Structure Identification: |
| Zhongguo Zhong Yao Za Zhi. 2014 Oct;39(19):3777-81. | | Study on chemical constituents from cultivated Gynura nepalensis[Pubmed: 25612439] |
METHODS AND RESULTS:
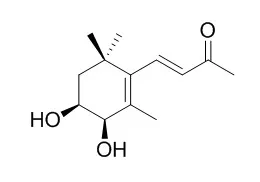
Taking application of some isolation and purification technologies, such as solvent extraction, preliminary solvent isolation, column chromatographies over silica gel and Sephadex LH-20 gel and preparative HPLC, 10 compounds were obtained from Gynura nepalensis cultivated in the suburban area of Beijing. Their structures were identified by spectroscopic methods and comparison with literature as (3R) -3-hydroxy-β-ionone (1), (3S,5R, 6S, 7E) -5, 6-epoxy-3-hydroxy-7-megastigmen-9-one (2), (+) -boscialin (3), 3, 6-trans-3-hydroxy-α-ionone (4), 3, 6-cis-3-hydroxy-α-ionone (5), 3, 4-cis-3,4-Dihydroxy-beta-ionone (6), ethyl caffeate (7), loliolide (8), 1H-indole-3-carbaldehyde (9), and 3-(hydroxyacetyl)indole (10), respectively. All compounds were isolated from the title plant for the first time, and with compounds 1, 2, 4-7, 9 and 10 being isolated from Gynura species for the first time. Structurally, the above compounds 1-6 belong to C13 nor-sesquiterpenoids, sharing the same carbon skeleton of megastigmane.
CONCLUSIONS:
According to this study, they are one of major kinds of chemical constituents of Gynura nepalensis and have important reference value for the investigation on phytotaxonomy of this species. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)