| In vitro: |
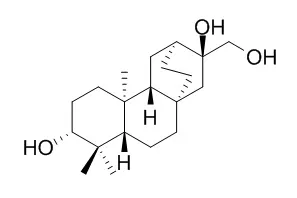
| Chinese Journal of Natural Medicines,2010,8(2):101-3. | | Diterpenes and triterpenes from the roots of Euphorbia fischeriana.[Reference: WebLink] |
To investigate the OFRs scavenging activities of homoisoflavonoids from the fibrous root of Ophiopogon japonicus.
METHODS AND RESULTS:
The scavenging effect of homoisoflavonoids isolated from O.japonicus extracts was examined on superoxide anion(O2·-),hydroxyl radical(·OH),and hydrogen peroxide(H2O2) by chemiluminescence.Ten homoisoflavonoids were obtained from O.japonicus extracts.They were identified as methylophiopogonanone A(1),methylophiopogonanone B(2),ophiopogonanone A(3),Ophiopogonanone E(4),5,7-dihydroxy-6,8-dimethyl-3-(4'-hydroxy-3',5'-methoxybenzyl) chroman-4-one(5),methylophiopogonone A(6),methylophiopogonone B(7),desmethylisoophiopogonone B(8),5,7,2'-trihydroxy-8-methyl-3-(3',4'-methylenedioxybenzyl) chromone(9) and 5,7,2'-trihydroxy-6,8-dimethyl-3-(3',4'-methylenedioxybenzyl) chromone(10).
CONCLUSIONS:
Compounds 8 and 9 were firstly isolated from O.japonicus.The majority of homoisoflavonoids could scavenge ·OH and H2O2 in vitro to a certain extent,and their bioactivities should be related with the respective structures. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)