| Description: |
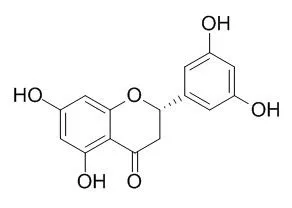
5,7,3',5'-Tetrahydroxyflavanone has anti-inflammatory properties, it can inhibit nitric oxide (NO) production with IC50 values of 18.5 uM, the inhibitory effect is accompanied by dose-dependent decreases in LPS-induced nitric oxide synthase (iNOS) in RAW 264.7 cells. It may have antiviral activity against HIV-1, it has inhibitory effects on HIV-1 reverse transcriptase (RT)-associated RNase H function. |
| In vitro: |
| Pathog Dis. 2013 Aug;68(3):116-24. | | Hypericum hircinum L. components as new single-molecule inhibitors of both HIV-1 reverse transcriptase-associated DNA polymerase and ribonuclease H activities.[Pubmed: 23821410 ] |
Among HIV-1 reverse transcriptase (RT)-associated functions, DNA polymerase and Ribonuclease H (RNase H) are both essential for HIV replication and excellent targets for drug development. While all RT inhibitors approved for therapy target the DNA polymerase activity, there is the pressing need for new RT inhibitors possibly targeting the RNase H function. In the last 20 years, many natural substances have shown antiviral activity against HIV-1, but only a few against the RNase H function.
METHODS AND RESULTS:
In this study, we have tested the ethanolic extracts obtained by the Hypericum hircinum L. (Hypericaceae) growing in Sardinia (Italy) on the HIV-1 RT-associated RNase H function and found that they have inhibitory effects. Active extracts were fractionated up to obtain the main components that have been isolated, tested, and identified to be betulinic acid, shikimic acid, chlorogenic acid, quercetin, 5,7,3',5'-tetrahydroxyflavanone(3',5,5',7-Tetrahydroxyflavanone), and 5,7,3',5'-tetrahydroxyflavanone 7-O-glucoside. Betulinic acid and 5,7,3',5'-tetrahydroxyflavanone 7-O-glucoside were active on both RT-associated activities, and betulinic acid was also active on HIV-1 mutant RTs resistant to efavirenz.
CONCLUSIONS:
Overall, our results suggest that some of these compounds inhibit the HIV-1 RT binding to an allosteric site previously described for other natural compounds and are potential leads for further drug development of a single molecules having dual inhibitory activity. | | Nat Prod Commun. 2015 Mar;10(3):383-5. | | Anti-inflammatory compounds from Ampelopsis cantoniensis.[Pubmed: 25924510] | Many natural products have been shown to have an inhibitory effect on nitric oxide (NO), and are used as chemotherapy agents for inflammation disease. The current study was designed to evaluate the anti-inflammatory activity of chemical components from the leaves of Ampelopsis cantoniensis.
METHODS AND RESULTS:
Sixteen compounds (1-16) were isolated and identified. Phloretin (5) and 5,7,3',5'-tetrahydroxyflavanone (3',5,5',7-Tetrahydroxyflavanone,16) inhibited nitric oxide (NO) production with IC50 values of 5.2, and 18.5 μM, respectively. The inhibitory effect of compounds 5 and 16 were accompanied by dose-dependent decreases in LPS-induced nitric oxide synthase (iNOS) in RAW 264.7 cells, respectively. This study investigated the significant anti-inflammatory properties of isolated compounds from the leaves of A. cantoniensis for the first time.
CONCLUSIONS:
The findings demonstrate that A. cantoniensis could be used beneficially in the treatment of inflammation disease. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)