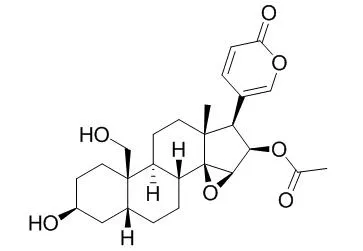
| Structure Identification: |
| Chemical Research in Chinese Universities, 2009, 25(6):801-806. | | Simultaneous Determination of Bufadienolides and Qualitative Evaluation for Venenum Bufonis by High Performance Liquid Chromatography[Reference: WebLink] |
A high performance liquid chromatographic method was used for the simultaneous identification and qua-litative evaluation of 12 bufadienolides(resibufogenin, cinobufagin, Cinobufaginol, arenobufagin, bufalin, bufotalin, gamabufotalin, cinobufotalin, Ψ-bufaranogin, desacetylcinobufagin, telocinobufagin and resibufogenol) in Venenum Bufonis.
METHODS AND RESULTS:
The chromatographic separation was performed on a Dikma C 18 analytical column via gradient elution with an aqueous solution of acetonitrile and 0.3% acetic acid at a flow rate of 0.8 mL/min. The method was validated to be acceptable in consideration of linearity(r 2 > 0.9992) and recovery(ranged from 98.9% to 102.0%). The limits of de-tection of the bufadienolides were from 0.48 ng for bufalin to 6.00 ng for cinobufotalin. The intra-day and inter-day precisions of the method were evaluated and were less than 3.0%. The method was successfully used to analyze 19 batches of Venenum Bufonis, and the similarity values between batches were calculated by Similarity Evaluation System for Chromatographic Fingerprint of TCM(Version 2004A, Chinese Pharmacopoeia Committee, Beijing).
CONCLUSIONS:
The results show that the contents of bufadienolides in the medicine and the similarity values based on these bufadieno-lides varied significantly from batch to batch. This proposed method could be utilized to qualify and control Venenum Bufonis to ensure its safety and efficacy in application. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)