| In vitro: |
| Bioorg Med Chem. 2006 Nov 15;14(22):7468-75. | | Phenylethanoid oligoglycosides and acylated oligosugars with vasorelaxant activity from Cistanche tubulosa.[Pubmed: 16908167 ] | The methanolic extract from the dried stems of Cistanche tubulosa (Schrenk) R. Wight was found to show an inhibitory effect on contractions induced by noradrenaline in isolated rat aortic strips.
METHODS AND RESULTS:
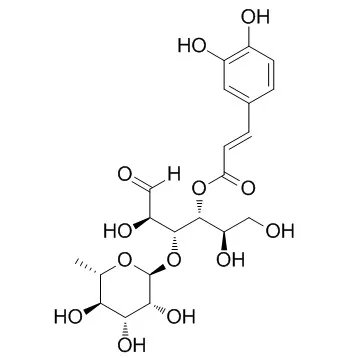
From the extract, new phenylethanoid oligoglycoside constituents, kankanosides F and G, and an acylated oligosugar, kankanose, were isolated together with 14 known compounds. The structures of these new compounds were determined on the basis of their chemical and physicochemical evidence. In addition, principal constituents, kankanoside F, kankanose, echinacoside, acteoside, and Cistanoside F, showed vasorelaxant activity, and several structural requirements for the activity were clarified. | | Biol Pharm Bull. 1996 Dec;19(12):1580-5. | | Antioxidative effects of phenylethanoids from Cistanche deserticola.[Pubmed: 8996643] |
The acetone-H2O (9:1) extract from the stem of Cistanche deserticola showed a strong free radical scavenging activity.
METHODS AND RESULTS:
Nine major phenylethanoid compounds were isolated from this extract. They were identified by NMR as acteoside, isoacteoside, 2'-acetylacteoside, tubuloside B, echinacoside, tubuloside A, syringalide A 3'-alpha-rhamnopyranoside, cistanoside A and Cistanoside F.
All of these compounds showed stronger free radical scavenging activities than alpha-tocopherol on 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical and xanthine/xanthine oxidase (XOD) generated superoxide anion radical (O2-.). Among the nine compounds, isoacteoside and tubuloside B, whose caffeoyl moiety is at 6'-position of the glucose, showed an inhibitory effect on XOD. We further studied the effects of these phenylethanoids on the lipid peroxidation in rat liver microsomes induced by enzymatic and non-enzymatic methods.
METHODS AND RESULTS:
As expected, each of them exhibited significant inhibition on both ascorbic acid/Fe2+ and ADP/NADPH/Fe3+ induced lipid peroxidation in rat liver microsomes, which were more potent than alpha-tocopherol of caffeic acid. The antioxidative effect was found to be potentiated by an increase in the number of phenolic hydroxyl groups in the molecule. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)