| In vitro: |
| J Agric Food Chem. 2003 Oct 22;51(22):6461-7. | | Comparison of antioxidative properties of carbazole alkaloids from Murraya koenigii leaves.[Pubmed: 14558763] |
METHODS AND RESULTS:
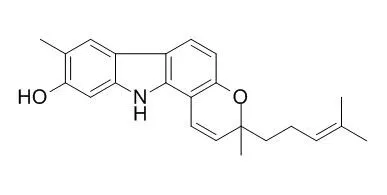
A new dimeric carbazole alkaloid, 8,10'-[3,3',11,11'-tetrahydro-9,9'-dihydroxy-3,3',5,8'-tetramethyl-3,3'-bis(4-methyl-3-pentenyl)]bipyrano[3,2-a]carbazole (12), was isolated from the CH(2)Cl(2) extract of Murraya koenigii together with six known carbazole alkaloids, koenimbine (6), O-methylmurrayamine A (7), O-methylmahanine (8), Isomahanine (9), bismahanine (10), and bispyrayafoline (11). Their structures were determined on the basis of (1)H and (13)C NMR spectroscopic and mass spectrometric (MS) data. The antioxidative properties of 12 carbazole alkaloids isolated from leaves of M. koenigii were evaluated on the basis of the oil stability index together with their radical scavenging ability against 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical. On the basis of the lag time to reach a steady state, the 12 carbazoles were classified into three groups.
CONCLUSIONS:
It is suggested that an aryl hydroxyl substituent on the carbazole rings plays a role in stabilizing the thermal oxidation and rate of reaction against DPPH radical. | | J. Agr. Chem. Env., 2013, 02(4):90-100. | | Antibacterial compounds from Rutaceae with activities against Flavobacterium columnare and Streptococcus iniae[Reference: WebLink] |
METHODS AND RESULTS:
From the ethyl acetate extract of Murraya koenegii (Rutaceae) leaves, Isomahanine (1) and mahanine (2) were isolated that showed antibacterial activity towards Flavobacterium columnare and Streptococcus iniae which caused columnaris disease and streptococcosis respectively. Isomahanine was found to have the strongest activity against F. columnare (isolate ALM-00-173) and S. iniae (isolate LA94-426) based on 24-h 50% inhibition concentration (IC50) and minimum inhibition concentration (MIC). Although compound (7), a nicotinamide isolated from Amyris texana had the lowest MIC (2.8 ± 0 mg/L) of any of the test compounds against F. columnare, the 24-h IC50 of 14.8 ± 0.6 mg/L was higher than that of Isomahanine and subsequently the 24-h IC50 RDC values for (7) were almost a magnitude of order higher than those obtained for Isomahanine.
CONCLUSIONS:
Isomahanine also had the strongest activity against S. iniae, with a 24-h IC50 of 1.3 ± 0.1 mg/L and MIC of 3.5 ± 0 mg/L, respectively. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)