| Kinase Assay: |
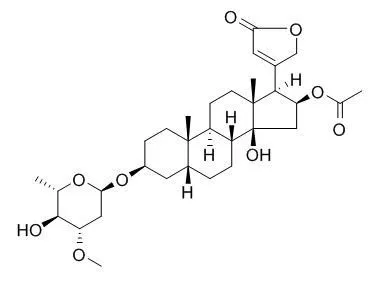
| Mol Carcinog. 2014 Apr;53(4):253-63. | | Cellular location and expression of Na+, K+ -ATPase α subunits affect the anti-proliferative activity of oleandrin.[Pubmed: 23073998] | The purpose of this study was to investigate whether intracellular distribution of Na(+), K(+) -ATPase α3 subunit, a receptor for cardiac glycosides including Oleandrin, is differentially altered in cancer versus normal cells and whether this altered distribution can be therapeutically targeted to inhibit cancer cell survival.
METHODS AND RESULTS:
The cellular distribution of Na(+), K(+) -ATPase α3 isoform was investigated in paired normal and cancerous mucosa biopsy samples from patients with lung and colorectal cancers by immunohistochemical staining. The effects of Oleandrin on α3 subunit intracellular distribution, cell death, proliferation, and EKR phosphorylation were examined in differentiated and undifferentiated human colon cancer CaCO-2 cells. While Na(+), K(+) -ATPase α3 isoform was predominantly located near the cytoplasmic membrane in normal human colon and lung epithelia, the expression of this subunit in their paired cancer epithelia was shifted to a peri-nuclear position in both a qualitative and quantitative manner. Similarly, distribution of α3 isoform was also shifted from a cytoplasmic membrane location in differentiated human colon cancer CaCO-2 cells to a peri-nuclear position in undifferentiated CaCO-2 cells. Intriguingly, Oleandrin exerted threefold stronger anti-proliferative activity in undifferentiated CaCO-2 cells (IC50, 8.25 nM) than in differentiated CaCO-2 cells (IC50, >25 nM). Oleandrin (10 to 20 nM) caused an autophagic cell death and altered ERK phosphorylation in undifferentiated but not in differentiated CaCO-2 cells.
CONCLUSIONS:
These data demonstrate that the intracellular location of Na(+), K(+) -ATPase α3 isoform is altered in human cancer versus normal cells. These changes in α3 cellular location and abundance may indicate a potential target of opportunity for cancer therapy. | | Biochem. Pharmacol.,2004, 66(11):2223-39. | | Oleandrin suppresses activation of nuclear transcription factor-κB and activator protein-1 and potentiates apoptosis induced by ceramide[Reference: WebLink] | Ceramide (N-acetyl-D-sphingosine), a second messenger for cell signaling induces transcription factors, like nuclear factor-kappa B (NF-kappa B), and activator protein-1 (AP-1) and is involved in inflammation and apoptosis. Agents that can suppress these transcription factors may be able to block tumorigenesis and inflammation. Oleandrin (trans-3,4',5-trihydroxystilbene), a polyphenolic cardiac glycoside derived from the leaves of Nerium oleander, has been used in the treatment of cardiac abnormalities in Russia and China for years.
METHODS AND RESULTS:
We investigated the effect of Oleandrin on NF-kappa B and AP-1 activation and apoptosis induced by ceramide. Oleandrin blocked ceramide-induced NF-kappa B activation. Oleandrin-mediated suppression of NF-kappa B was not restricted to human epithelial cells; it was also observed in human lymphoid, insect, and murine macrophage cells. The suppression of NF-kappa B coincided with suppression of AP-1. Ceramide-induced reactive intermediates generation, lipid peroxidation, cytotoxicity, caspase activation, and DNA fragmentation were potentiated by Oleandrin. Oleandrin did not show its activity in primary cells.
CONCLUSIONS:
Oleandrin's anticarcinogenic, anti-inflammatory, and growth-modulatory effects may thus be partially ascribed to the inhibition of activation of NF-kappa B and AP-1 and potentiation of apoptosis. | | Integr Cancer Ther. 2007 Dec;6(4):354-64. | | Autophagic cell death of human pancreatic tumor cells mediated by oleandrin, a lipid-soluble cardiac glycoside.[Pubmed: 18048883] | Lipid-soluble cardiac glycosides such as bufalin, Oleandrin, and digitoxin have been suggested as potent agents that might be useful as anticancer agents. Past research with Oleandrin, a principle cardiac glycoside in Nerium oleander L. (Apocynaceae), has been shown to induce cell death through induction of apoptosis.
METHODS AND RESULTS:
In PANC-1 cells, a human pancreatic cancer cell line, cell death occurs not through apoptosis but rather through autophagy. Oleandrin at low nanomolar concentrations potently inhibited cell proliferation associated with induction of a profound G(2)/M cell cycle arrest. Inhibition of cell cycle was not accompanied by any significant sub G1 accumulation of cells, suggesting a nonapoptotic mechanism. Oleandrin-treated cells exhibited time- and concentration-dependent staining with acridine orange, a lysosomal stain. Subcellular changes within PANC-1 cells included mitochondrial condensation and translocation to a perinuclear position accompanied by vacuoles. Use of a fluorescent Oleandrin analog (BODIPY-Oleandrin) revealed co-localization of the drug within cell mitochondria. Damaged mitochondria were found within autophagosome structures. Formation of autophagosomes was confirmed through electron microscopy and detection of green fluorescent protein-labeled light chain 3 association with autophagosome membranes. Also observed was a drug-mediated inhibition of pAkt formation and up-regulation of pERK. Transfection of Akt into PANC-1 cells or inhibition of pERK activation by MAPK inhibitor abrogated Oleandrin-mediated inhibition of cell growth, suggesting that the reduction of pAkt and increased pERK are important to Oleandrin's ability to inhibit tumor cell proliferation.
CONCLUSIONS:
The data provide insight into the mechanisms and role of a potent, lipid-soluble cardiac glycoside (Oleandrin) in control of human pancreatic cancer proliferation. |
|
| Cell Research: |
| Br J Pharmacol. 2014 Jul;171(14):3339-51. | | Short-term exposure to oleandrin enhances responses to IL-8 by increasing cell surface IL-8 receptors.[Pubmed: 24172227] | One of the first steps in host defence is the migration of leukocytes. IL-8 and its receptors are a chemokine system essential to such migration. Up-regulation of these receptors would be a viable strategy to treat dysfunctional host defence. Here, we studied the effects of the plant glycoside Oleandrin on responses to IL-8 in a human monocytic cell line.
METHODS AND RESULTS:
U937 cells were incubated with Oleandrin (1-200 ng mL(-1) ) for either 1 h (pulse) or for 24 h (non-pulse). Apoptosis; activation of NF-κB, AP-1 and NFAT; calcineurin activity and IL-8 receptors (CXCR1 and CXCR2) were measured using Western blotting, RT-PCR and reporter gene assays.
Pulse exposure to Oleandrin did not induce apoptosis or cytoxicity as observed after non-pulse exposure. Pulse exposure enhanced activation of NF-κB induced by IL-8 but not that induced by TNF-α, IL-1, EGF or LPS. Exposure to other apoptosis-inducing compounds (azadirachtin, resveratrol, thiadiazolidine, or benzofuran) did not enhance activation of NF-κB. Pulse exposure to Oleandrin increased expression of IL-8 receptors and chemotaxis, release of enzymes and activation of NF-κB, NFAT and AP-1 along with increased IL-8-mediated calcineurin activation, and wound healing. Pulse exposure increased numbers of cell surface IL-8 receptors.
CONCLUSIONS:
Short-term (1 h; pulse) exposure to a toxic glycoside Oleandrin, enhanced biological responses to IL-8 in monocytic cells, without cytoxicity. Pulse exposure to Oleandrin could provide a viable therapy for those conditions where leukocyte migration is defective. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)