| Kinase Assay: |
| Biochem Biophys Res Commun. 2004 Oct 15;323(2):668-73. | | Influence of lipophilicity on the interactions of hydroxy stilbenes with cytochrome P450 3A4.[Pubmed: 15369802 ] | Resveratrol, a polyphenol found in red wine, was recently suggested to act as an irreversible, mechanism-based inactivator of cytochrome P450 3A4 (CYP3A4).
METHODS AND RESULTS:
We found a significant inhibition of human CYP3A4-dependent transformation of cyclosporine by resveratrol, with IC50 = 4.5 microM. We studied the kinetics parameters of CYP3A4 transformation of resveratrol and structurally related, naturally occurring stilbenes. Resveratrol, piceid, Resveratroloside, 5,4'-dihydroxy-3-O-methoxystilbene, and 5,3-dihydroxy-4'-O-methoxystilbene were all shown to inhibit hydroxylation of testosterone by CYP3A4. Both methoxy-stilbenes had lower IC50 values, ranging from 0.43 to 0.47 microM, suggesting that lipophilicity rather than number or positions of free hydroxyls (3,5 or 5,4') determines the CYP3A4 inhibition capacity of polyphenols. In line with these findings, both glucosyl-stilbenes were found to be weak inhibitors of CYP3A4. The affinity of the enzyme towards methoxy-stilbenes, expressed as apparent Km, was indeed higher than those for the parent resveratrol and its glucosides, in CYP3A4 reaction mixtures. Vmax values were similar, except for piceid.
CONCLUSIONS:
These results support the role of lipophilicity in the interaction of polyphenols with CYP3A4. It is suggested that selective structural modifications of substrates add significantly to knowledge acquired through molecular modifications of the enzyme. | | J Nutr Sci Vitaminol (Tokyo). 2012;58(4):278-86. | | Analysis and functionality of major polyphenolic components of Polygonum cuspidatum (itadori).[Pubmed: 23132312] | Polygonum cuspidatum has been broadly utilized as a herbal medicine in Asia, but the outline of the polyphenol compounds in the plant has not been characterized well.
METHODS AND RESULTS:
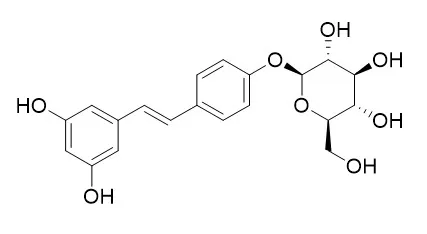
In the present study, the major polyphenolic components were isolated from the roots of P. cuspidatum, and identified as resveratrol and its glucosides, Resveratroloside and polydatin. On the other hand, chlorogenic acid was found to be one of the polyphenolic components in the leaves and stems of the plant. Next, we elucidated that resveratrol derivatives and chlorogenic acid exhibit α-glucosidase inhibitory activities, and Resveratroloside exhibits the same inhibitory activity as quercetin does.
Furthermore, DPPH radical scavenging activities of antioxidants including resveratrol derivatives and chlorogenic acid derivatives were examined by initial rate analyses of their reactions.
Subsequently, it was revealed that resveratrol derivatives have slow-acting effects on the radical scavenging activity and that chlorogenic acid derivatives exhibit very fast-acting effects. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)