| In vitro: |
| South African Journal of Botany,2017,111:93-8. | | Antioxidant and free radical scavenging activities of taxoquinone, a diterpenoid isolated from Metasequoia glyptostroboides[Reference: WebLink] |
METHODS AND RESULTS:
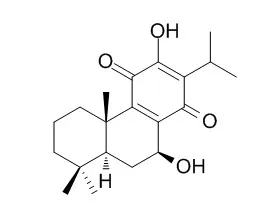
An abietane type diterpenoid Taxoquinone, isolated from ethyl acetate cone extract of Metasequoia glyptostroboides, was analyzed for its antioxidant efficacy as reducing power ability and its ability to scavenge free radicals such as 1,1-diphenyl-2-picryl hydrazyl (DPPH), nitric oxide, superoxide and hydroxyl radicals. As a result, Taxoquinone showed significant and concentration-dependent antioxidant and free radical scavenging activities of DPPH, nitric oxide, superoxide and hydroxyl free radicals by 78.83%, 72.42%, 72.99% and 85.04%, as compared to standard compounds ascorbic acid (81.69%, 74.62%, 73% and 73.79%) and α-tocopherol/butylated hydroxyanisole (84.09%, 78.61%, 74.45% and 70.02%), respectively.
CONCLUSIONS:
These findings justify the biological and traditional uses of M. glyptostroboides or its secondary metabolite Taxoquinone as confirmed by its promising antioxidant and free radical scavenging activities. | | Bangladesh Journal of Pharmacology,2014,9(2):154-60. | | Antimycotic potential of a diterpenoid taxoquinone against Candida species isolated from Metasequoia glyptostroboides[Reference: WebLink] | The objective of this study was to confirm the antimycotic potential of a diterpenoid compound Taxoquinone, isolated from Metasequoia glyptostroboides against pathogenic isolates of Candida species.
METHODS AND RESULTS:
The Taxoquinone (100 μg/disc) displayed potential anticandidal effect against Candia albicans KBN06P00076, KBN06P00074, C. glabrata KBN06P00066, KBN06P00068, C. tropicalis KBN06P00682, KBN06P00058, C. parapsilosis KBN06P00060, KBN06P00055, and C. guilliermondii KBN06P00492 and KBN06P00867 as a diameter of zones of inhibition, found in the range of 10 ± 0.6 to 13 ± 1.1 mm. The minimum inhibitory and minimum fungicidal concentrations of Taxoquinone against the tested clinical isolates were found in the range of 125 to 500 and 125 to 1,000 μg/mL, respectively.
On the viable counts of the tested fungal isolates, the Taxoquinone evoked a remarkable anticandidal effect.
METHODS AND RESULTS:
Elaborative study of SEM revealed potential detrimental effect of Taxoquinone on the morphology of C. parapsilosis KBN06P00060 at MIC concentration. These findings confirmed therapeutic potential of Taxoquinone. | | BMC Complement Altern Med. 2015 Mar 26;15:84. | | α-Glucosidase and tyrosinase inhibitory effects of an abietane type diterpenoid taxoquinone from Metasequoia glyptostroboides[Pubmed: 25887244 ] |
METHODS AND RESULTS:
In this study, a diterpenoid compound Taxoquinone, isolated from Metasequoia glyptostroboides was evaluated for its α-glucosidase and tyrosinase inhibitory efficacy in terms of its potent anti-diabetic and depigmentation potential, respectively.:As a result, Taxoquinone at the concentration range of 100-3,000 μg/mL and 200-1,000 μg/mL showed potent efficacy on inhibiting α-glucosidase and tyrosinase enzymes by 9.24-51.32% and 11.14-52.32%, respectively.
CONCLUSIONS:
The findings of this study clearly evident potent therapeutic efficacy of an abietane diterpenoid Taxoquinone isolated from M. glyptostroboides with a possibility for using it as a novel candidate in food and medicine industry as a natural alternative medicine to prevent diabetes mellitus type-2 related disorders and as a depigmentation agent. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)