| Kinase Assay: |
| Free Radic Biol Med. 2015 Jan;78:135-46. | | Identification of heme oxygenase-1 stimulators by a convenient ELISA-based bilirubin quantification assay.[Pubmed: 25462643 ] | The upregulation of heme oxygenase-1 (HO-1) has proven to be a useful tool for fighting inflammation. In order to identify new HO-1 inducers, an efficient screening method was developed which can provide new lead structures for drug research.
METHODS AND RESULTS:
We designed a simple ELISA-based HO-1 enzyme activity assay, which allows for the screening of 12 compounds in parallel in the setting of a 96-well plate. The activity of known HO-1 inducers was confirmed, while the chalcones cardamonin, flavokawain A, calythropsin, 2',3,4'-trihydroxy-4-methoxychalcone (4-O-Methylbutein,THMC), and 2',4'-dihydroxy-3,4-dimethoxychalcone (DHDMC) were identified as new potent HO-1 inducers. The highest inductive power after 6h incubation was found at 10 uM for DHDMC (6.1-fold), carnosol (3.9-fold), butein (3.1-fold), THMC (2.9-fold), and zerumbone (2.5-fold). Moreover, the time dependence of HO-1 protein production for DHDMC was compared to its enzyme activity, which was further evaluated in the presence of lipopolysaccharide and the specific HO-1 inhibitor tin protoporphyrin IX.
CONCLUSIONS:
Taken together, we developed a convenient and highly sensitive ELISA-based HO-1 enzyme activity assay, allowing the identification and characterization of molecules potentially useful for the treatment of inflammatory and autoimmune diseases. |
|
| Structure Identification: |
| Phytochemistry,1975,14(7):1621-3. | | Flavonoids and polyacetylenes in Dahlia tenuicaulis[Reference: WebLink] |
METHODS AND RESULTS:
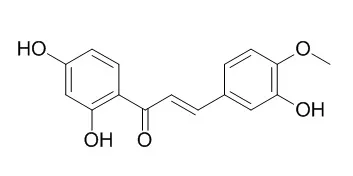
5,7,4′-Trimethoxyflavanone,5-hydroxy-7,4′-dimethoxyflavanone,2′-hydroxy-4,4′,6′-trimeth-oxychalcone, and a new naturally occurring compound, 5,7,4′-trimethoxyflavan-4-ol have been isolated from the leaves of Dahlia tenuicaulis Sorensen. Two chalcones, 4,2′,4′-trihydroxychalcone and 3,2′,4′-trihydroxy-4-methoxychalcone(4-O-Methylbutein), and 5-hydroxy-7,4′-dimethoxyflavanone have been isolated from the flower heads.
CONCLUSIONS:
Minute amounts of the polyacetylene 1,3-diacetoxy-tetradeca-4,6-diene-8,10,12-triyne have been found in both leaves and flower heads, whereas 1-acetoxy-tetradeca-4,6-diene-8,10,12-triyne was present only in the flower heads. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)