| In vitro: |
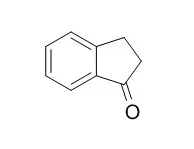
| Eur J Pharm Sci. 2012 Oct 9;47(3):596-603. | | Antiviral activity against the hepatitis C virus (HCV) of 1-indanone thiosemicarbazones and their inclusion complexes with hydroxypropyl-β-cyclodextrin.[Pubmed: 22885176 ] | The hepatitis C virus (HCV) is a major cause of acute and chronic hepatitis in humans. Approximately 5% of the infected people die from cirrhosis or hepatocellular carcinoma. The current standard therapy comprises a combination of pegylated-interferon alpha and ribavirin. Due to the relatively low effectiveness, the prohibitive costs and the extensive side effects of the treatment, an intense research for new direct-acting anti-HCV agents is taking place. Furthermore, NS3 protease inhibitors recently introduced into the market are not effective against all HCV subgenotypes. Thiosemicarbazones (TSCs) have shown antiviral activity against a wide range of DNA and RNA viruses. However, their extremely low aqueous solubility and high self-aggregation tendency often preclude their reliable biological evaluation in vitro.
METHODS AND RESULTS:
In this work, we investigated and compared for the first time the anti-HCV activity of two 1-Indanone TSCs, namely 5,6-dimethoxy-1-Indanone TSC and 5,6-dimethoxy-1-Indanone N4-allyl TSC, and their inclusion complexes with hydroxypropyl-β-cyclodextrin (HPβ-CD) in Huh-7.5 cells containing the full-length and the subgenomic subgenotype 1b HCV replicon system. Studies of physical stability in culture medium showed that free TSCs precipitated rapidly and formed submicron aggregates. Conversely, TSC complexation with HPβ-CD led to more stable systems with minimal size growth and drug concentration loss. More importantly, both TSCs and their inclusion complexes displayed a potent suppression of the HCV replication in both cell lines with no cytotoxic effects.
CONCLUSIONS:
The mechanism likely involves the inhibition of non-structural proteins of the virus. In addition, findings suggested that the cyclodextrin released the drug to the culture medium over time. This platform could be exploited for the study of the drug toxicity and pharmacokinetics animal models. | | J Microbiol Biotechnol. 2012 Jun;22(6):832-7. | | Oxidative potential of some endophytic fungi using 1-indanone as a substrate.[Pubmed: 22573162] |
METHODS AND RESULTS:
The oxidative potential of the fungus Penicillium brasilianum, a strain isolated as an endophyte from a Meliaceae plant (Melia azedarach), was investigated using 1-Indanone as a substrate to track the production of monooxygenases. The fungus produced the dihydrocoumarin from 1-Indanone with the classical Baeyer-Villiger reaction regiochemistry, and (-)-(R)-3-hydroxy-1-Indanone with 78% ee. Minor compounds resulting from lipase and SAM activities were also detected. The biotransformation procedures were also applied to a collection of Penicillium and Aspergillus fungi obtained from M. azedarach and Murraya paniculata.
CONCLUSIONS:
The results showed that Baeyer-Villiger were mostly active in fungi isolated from M. azedarach. Almost all of the fungi tested produced 3-hydroxy-1-Indanone. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)