To determine the effect of oral dosing of sheep with Loline alkaloids on their excretion in urine and faeces, and to monitor for any toxic effects.
METHODS AND RESULTS:
In Experiment 1, six 9-month-old ewe lambs were given a single oral dose of Loline alkaloids (52 mg/kg bodyweight (BW); acute exposure) as a suspension of ground meadow fescue (Festuca pratensis) seed in water. In Experiment 2, on six consecutive days, six ewe lambs were given three doses of Loline (68 mg/kg BW/day; chronic exposure). Blood was collected at variable intervals up to 72 h in Experiment 1, and up to 8 days in Experiment 2, for haematology and measurement of alkaline phosphatase, aspartate aminotransaminase, creatine kinase and γ-glutamyl transferase in plasma. Urine and faecal samples were collected at similar times for measurement of creatinine in urine and Loline alkaloid analysis. A post mortem with histopathology was carried out on two animals at the end of each experiment.
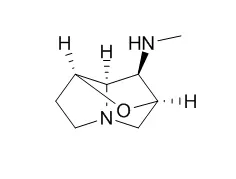
The Loline alkaloids, N-acetyl norLoline, N-formyl Loline, N-acetyl Loline, N-methyl Loline and Loline base were detected in urine within 15 minutes after the single dosing. N-formyl Loline and Loline base were the predominant metabolites in urine in both experiments. The total quantity of Lolines excreted in both urine and faeces was 10% and 4% of the amount dosed in Experiments 1 and 2, respectively. In both experiments, the clinical chemistry parameters in blood and urine were within normal ranges. Post-mortem and histopathological examination did not show any abnormalities.
CONCLUSIONS:
This is the first report of Loline alkaloid profiles in both urine and faeces of sheep. The appearance of Loline alkaloids and the Loline base in urine within 15 minutes suggests rapid uptake, metabolism and excretion. Loline alkaloids were non-toxic to sheep at the concentrations they are exposed to under New Zealand grazing conditions. The low recovery of Loline alkaloids in urine and faeces in the absence of toxicity signs suggests Lolines are extensively metabolised; probably to forms other than N-formyl Loline, N-methyl Loline, N-acetyl Loline, N-acetyl norLoline, and Loline base in the digestive tract of sheep prior to absorption, and/or in the liver or other tissues following absorption. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)