METHODS AND RESULTS:
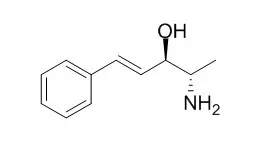
Chiral resolution of a number of cationic drugs of forensic interest (amphetamine, methamphetamine, cathinone, methcathinone, cathine, cocaine, propoxyphene, and various alpha-hydroxyphenethylamines) is achieved via capillary electrophoresis (CE) with added cyclodextrins (CDs), including novel mixtures of neutral and anionic CDs. In the latter studies, resolution and migration speed are readily adjusted by varying the ratio of the two added CDs, as the anionic CD acts as a counter-migrating complexing reagent. The neutral CD, heptakis(2,6-di-O-methyl)-beta-CD, was found suitable for the analysis of illicit cocaine and khat leaves (Catha edulis Forsk), which contain (-)-alpha-aminopropiophenone ((-)-cathinone), (+)-norpseudoephedrine (cathine), (-)-norephedrine, and trace levels of the phenylpentenylamines (+)-merucathinone, (+)-Merucathine, and possibly (-)-pseudoMerucathine.
CONCLUSIONS:
The use of mixtures of the neutral and the anionic CD (beta-CD sulfobutyl ether IV) was found suitable for the analysis of illicit amphetamine, methamphetamine, methcathinone, and propoxyphene. A model is presented for the impact of mixtures of neutral and anionic CDs on migration behavior and chiral resolution in CE. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)