| Structure Identification: |
| Phytochemistry. 2002 Mar;59(5):537-42. | | Non-glycosidic iridoids from Cymbaria mongolica.[Pubmed: 11853749] |
METHODS AND RESULTS:
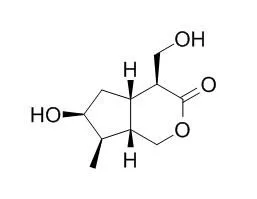
Six non-glycosidic iridoids, i.e. (1R,4S,4aS,7S,7aS)-7-hydroxyl-4-hydroxy- methyl-7-methyl-1-methoxyl-1,4,4a,7a-tetrahydrocyclopenta[e]pyran-3-one (1), (1S,4R,4aS,7S,7aS)-7-hydroxyl-4-hydroxymethyl-7-methyl-1-methoxyl-1,4,4a,7a-tetrahydrocyclopenta[e]pyran-3-one (2), (1R,4R,4aS,7S,7aS)-7-hydroxyl-4-hydroxy-methyl-7-methyl-1-methoxyl-1,4,4a,7a-tetrahydrocyclopenta[e]pyran-3-one (3), (1R, 4R, 4aS, 7aS)-4,7-dihydroxymethyl-1-methoxyl-1,4,4a,7a-tetrahydrocyclopenta-6-ene[e]pyran-3-one (4), (1R, 4R, 4aS, 7aS)-4,7-dihydroxymethyl-1-hydroxyl-1,4,4a, 7a-tetrahydrocyclopenta-6-ene[e]pyran-3-one (5), (1R, 4S, 4aS, 7aS)-4,7-dihydroxy-methyl-1-methoxyl-1,4,4a,7a-tetrahydrocyclopenta-6-ene[e]pyran-3-one (6), as well as five known non-glycosidic iridoids mussaenin A (7), gardendiol (8), isoboonein (9), 4-Epialyxialactone (10) and rehmaglutin D (11) have been isolated from the Chinese medicinal plant Cymbaria mongolica.
CONCLUSIONS:
Their structures were elucidated by spectroscopic methods. These compounds exhibit significant antitumor and antibacterial activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)