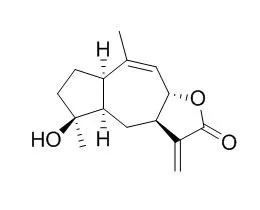
| Structure Identification: |
| Chem Pharm Bull (Tokyo). 2008 Nov;56(11):1535-45. | | New eudesmane derivatives and other sesquiterpenes from the epigeal parts of Dittrichia graveolens.[Pubmed: 18981602] |
METHODS AND RESULTS:
In bioassay-guided searches for novel bioactive natural products from higher plants of the Egyptian flora, two new eudesmane sesquiterpene derivatives, 3alpha-hydroxyilicic acid methyl ester (1) and 2alpha-hydroxy-4-epi-ilicic acid (2), together with 11 known sesquiterpenes were isolated from bioactive fractions of the active epigeal parts extracts of Dittrichia graveolens (L.) GREUTER (Asteraceae) growing in the coastal regions of northwestern Egypt. Four other known sesquiterpene lactones with different carbon skeletons, named 2alpha-hydroxy-2R-xanthalongin (8), 4-Epi-isoinuviscolide (9), 8-epi-helenalin (10), and bigelovin (11), were also isolated for the first time from the same source. The stereochemical structures of the isolated compounds were elucidated on the basis of physical and spectroscopic methods including UV, IR, 1H-, 13C-NMR, distortionless enhancement by polarization transfer, 2D NMR, 1H-1H correlation spectroscopy, 1H-13C heteronuclear single-quantum coherence, 1H-13C heteronuclear multiple-bond connectivity, 1H-1H nuclear Overhauser effect spectroscopy experiments, and high-resolution mass spectrometry, as well as some chemical transformations.
CONCLUSIONS:
The antimicrobial, antiinflammatory, and antipyretic activities of D. graveolens extracts and chromatographic fractions were carried out and the various bioactivities of our findings are discussed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)