| J Gastrointest Oncol. 2015 Jun;6(3):E40-3. |
| Capecitabine-induced leukocytoclastic vasculitis under neoadjuvant chemotherapy for locally advanced colorectal cancer.[Pubmed: 26029464] |
We describe a case of Capecitabine-induced leukocytoclastic vasculitis in a patient with locally advanced rectal cancer under curative neoadjuvant concurrent chemoradiation using Capecitabine.
METHODS AND RESULTS:
After 5 days of the initiation of Capecitabine the patient developed a pruritic maculopapular rash in her extremities consistent with vasculitis which was confirmed on skin biopsy without any signs of systemic involvement. Capecitabine was held and the rash was treated with topical steroids with complete resolution of both rash and pruritus. Due to a lack of other alternative chemotherapeutic options and the cutaneous-only involvement of vasculitis, the Capecitabine was re-introduced. Two days later, the patient developed an identical maculopapular rash with a similar distribution. Prednisone was initiated while the Capecitabine was continued with complete resolution of the rash. The patient successfully completed her curative neoadjuvant chemoradiation therapy treatment without the need to permanently discontinue the Capecitabine. |
| Curr Med Res Opin. 2015 Apr;31(4):731-41. |
| Observational study of adjuvant therapy with capecitabine in colon cancer.[Pubmed: 25651480] |
This observational study was conducted to document the safety of Capecitabine-based adjuvant therapy in patients with resected colon cancer under routine clinical conditions.
METHODS AND RESULTS:
ML20431 was a prospective, multicenter, non-interventional, observational study. It was designed to answer five research questions relating to safety, dosage and administration, and discontinuation from Capecitabine-based adjuvant therapy. Patients were required to have R0 resected stage III colon cancer and have started treatment with Capecitabine-based adjuvant therapy based on a decision by the investigator. Patients were followed over an observation period of ≤6 months after initiation of therapy. Investigators were required to complete the study case report form at study entry, each treatment cycle, and at the final examination. A total of 1485 patients were included in the study, and 1481 patients were treated with Capecitabine and formed the analysis population. Most patients had colon cancer (78.3%), followed by rectal cancer (16.4%). Most patients had stage III disease (69.3%); the remaining patients had stage II disease (30.7%). The most common all-grade adverse reactions were hand-foot syndrome (46.9%), diarrhea (34.4%), and hemoglobin decreases (31.5%). Grade 3/4 adverse reactions were infrequent (<4%). Serious adverse events were reported in 96 patients (6.5%). Six or more cycles of treatment were completed by 77.9% of patients. Approximately two-thirds of patients (67.3%) received Capecitabine monotherapy and the remainder (32.7%) received Capecitabine in combination with ≥1 drugs, most commonly oxaliplatin (460 cases). Discontinuation of Capecitabine was documented in 344 patients (23.2%). no efficacy data were collected; the questionnaires for patients' expectations and satisfaction were not formally validated; and a few patients (<1.5%) had some retrospective data.
CONCLUSIONS:
The safety profile of Capecitabine-based adjuvant therapy in a broad patient population with colon cancer is similar to that previously documented in phase III clinical trials. |
| Anticancer Res. 2014 Dec;34(12):7239-45. |
| Concomitant capecitabine with hepatic delivery of drug eluting beads in metastatic colorectal cancer.[Pubmed: 25503155] |
Effectiveness and toxicity of transcatheter arterial injection of irinotecan-eluting beads (DEBIRI) with and without concurrent Capecitabine in pre-treated patients with metastatic colorectal cancer (CRC).
METHODS AND RESULTS:
An Institutional Review Board-approved, multi-institutional registry from 5/2008 to 8/2013 was reviewed. Patients who received DEBIRI with (X-DEBIRI) or without (DEBIRI) Capecitabine were compared. Twenty-two X-DEBIRI and 149 DEBIRI patients were compared. There was no difference in the two groups with regards to adverse events (p=0.56). During a 3- and 6-month evaluation, the disease control rate (DCR) was similar in both groups. During the 12-month evaluation, there was better DCR in the X-DEBIRI group (p=0.03). Median survival was 13 months in the DEBIRI group and 22 months in the X-DEBIRI group (log-rank test, p=0.217).
CONCLUSIONS:
There is no additional toxicity when adding Capecitabine with DEBIRI. Concurrent Capecitabine may offer more durable disease control rate compared to DEBIRI-alone. Survival benefit with concurrent Capecitabine was not statistically significant but there may be a trend towards improved survival. |


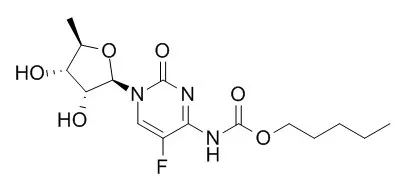



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)