| In vitro: |
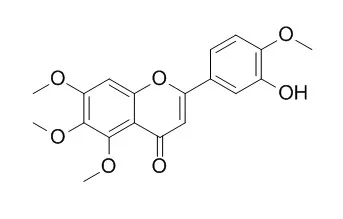
| Toxicology Letters, 2014, 230(2):304-313. | | Benzo[a]pyrene sensitizes MCF7 breast cancer cells to induction of G1 arrest by the natural flavonoid eupatorin-5-methyl ether, via activation of cell signaling proteins and CYP1-mediated metabolism.[Reference: WebLink] | Eupatorin-5-methylether (E5M) is a flavone containing 4 methoxy groups that is present in plants with medicinal activity, whereas luteolin (L) is a polyhydroxylated flavone commonly encountered in dietary products. In the present study we investigated the interaction of the two flavonoids with cytochrome P450 CYP1 enzymes in breast cancer MCF7 cells.
METHODS AND RESULTS:
Both compounds induced a dose dependent increase in CYP1A1 and CYP1B1 mRNA levels, as well as in EROD activity, a marker of CYP1 enzyme activity. Induction of cytochrome P450 CYP1 expression by E5M was accompanied by translocation of the ligand-activated transcription factor AhR to the nucleus, as demonstrated by confocal immunofluoresence. More importantly, although E5M was less active than L in inhibiting proliferation of MCF7 cells, when the cells were pretreated with the CYP1 inducer Benzo[a]pyrene (BaP) the potency of E5M was augmented. HPLC and LC-MS analysis revealed that E5M was metabolized to a major conversion product assigned E5M1 resulting from one step demethylation reaction in MCF7 cells whereas L metabolism by recombinant CYP1A1 did not reveal any metabolites. E5M1 production in BaP-induced MCF7 cells was attenuated in the presence of the CYP1A1 inhibitor α-napthoflavone. E5M further induced a dose dependent increase in the cell signaling proteins p21, JNK and p-JNK in MCF7 cells. This effect was enhanced in BaP pretreated cells and was associated with G1 arrest and a small percentage of apoptosis (3.5%). E5M antiproliferative effect in BaP pretreated cells was attenuated in the presence of the CYP1A1 inhibitor α-napthoflavone, as demonstrated by Western blotting and FACS analysis.
CONCLUSIONS:
Taken together the results demonstrate that BaP sensitizes MCF7 cells to E5M antiproliferative activity via enhanced induction of p21, JNK and p-JNK that in turn results by cytochrome P450 CYP1-mediated conversion to the metabolite E5M1. | | Planta Medica, 2012, 78(08):779-786. | | Flavonoids eupatorin and sinensetin present in Orthosiphon stamineus leaves inhibit inflammatory gene expression and STAT1 activation.[Reference: WebLink] | Cytokines and other inflammatory mediators, such as prostaglandin E₂ (PGE₂) and nitric oxide (NO) produced by cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), respectively, activate and drive inflammation and therefore serve as targets for anti-inflammatory drug development. Orthosiphon stamineus is an indigenous medicinal plant of Southeast Asia that has been traditionally used in the treatment of rheumatoid arthritis, gout, and other inflammatory disorders.
METHODS AND RESULTS:
The present study investigated the anti-inflammatory properties of Orthosiphon stamineus leaf chloroform extract (CE), its flavonoid-containing CE fraction 2 (CF2), and the flavonoids eupatorin, Eupatorin-5-methylether (TMF), and sinensetin, identified from the CF2. It was found that CE (20 and 50 µg/mL) and CF2 (20 and 50 µg/mL) inhibited iNOS expression and NO production, as well as PGE₂ production. Eupatorin and sinensetin inhibited iNOS and COX-2 expression and the production of NO (IC₅₀ 5.2 µM and 9.2 µM for eupatorin and sinensetin, respectively) and PGE₂ (IC₅₀ 5.0 µM and 2.7 µM for eupatorin and sinensetin, respectively) in a dose-dependent manner. The extracts and the compounds also inhibited tumor necrosis factor α (TNF-α) production (IC₅₀ 5.0 µM and 2.7 µM for eupatorin and sinensetin, respectively). Eupatorin and sinensetin inhibited lipopolysaccharide (LPS)-induced activation of transcription factor signal transducers and activators of transcription 1α (STAT1α). Furthermore, eupatorin (50 mg/kg i. p.) and sinensetin (50 mg/kg i. p.) inhibited carrageenan-induced paw inflammation in mice.
CONCLUSIONS:
The results suggest that CE and CF2, as well as the known constituents of CF2, i.e., eupatorin and sinensetin, have meaningful anti-inflammatory properties which may be utilized in the development of novel anti-inflammatory treatments. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)