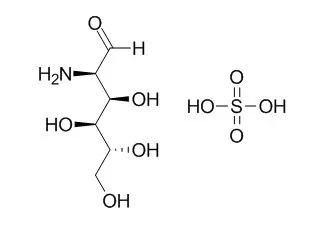
| Description: |
Glucosamine sulfate has environmental antibacterial activity. Glucosamine sulfate could as a safe symptomatic Slow Acting Drug for osteoarthritis, it can stimulate proteoglycan synthesis by chondrocytes and has mild anti-inflammatory properties, it is therefore as effective as ibuprofen on symptoms of knee osteoarthritis. |
| Targets: |
IL Receptor | MMP(e.g.TIMP) | Antifection |
| In vitro: |
| Spine J. 2013 Dec 18. pii: S1529-9430(13)01987-6 | | The effects of glucosamine sulfate on intervertebral disc annulus fibrosus cells in vitro.[Pubmed: 24361347] | Glucosamine has gained widespread use among patients, despite inconclusive efficacy data. Inconsistency in the clinical literature may be related to lack of understanding of the effects of glucosamine on the intervertebral disc, and therefore, improper patient selection.
The goal of our study was to investigate the effects of glucosamine on intervertebral disc cells in vitro under the physiological conditions of inflammation and mechanical loading.
Controlled in vitro laboratory setting.
METHODS AND RESULTS:
Intervertebral disc cells isolated from the rabbit annulus fibrosus were exposed to Glucosamine sulfate in the presence and absence of interleukin-1β and tensile strain. Outcome measures included gene expression, measurement of total glycosaminoglycans, new proteoglycan synthesis, prostaglandin E2 production, and matrix metalloproteinase activity. The study was funded by NIH/NCCAM, and the authors have no conflicts of interest.
Under conditions of inflammatory stimulation alone, glucosamine demonstrated a dose-dependent effect in decreasing inflammatory and catabolic mediators and increasing anabolic genes. However, under conditions of mechanical stimulation, although inflammatory gene expression was decreased, PGE2 was not. In addition, matrix metalloproteinase-3 gene expression was increased and aggrecan expression decreased, both of which would have a detrimental effect on matrix homeostasis. Consistent with this, measurement of total glycosaminoglycans and new proteoglycan synthesis demonstrated detrimental effects of glucosamine under all conditions tested.
CONCLUSIONS:
These results may in part help to explain the conflicting reports of efficacy, as there is biological plausibility for a therapeutic effect under conditions of predominate inflammation but not under conditions where mechanical loading is present or in which matrix synthesis is needed. | | Clin Rheumatol. 2009 Oct;28(10):1221-3. | | Glucosamine sulfate--environmental antibacterial activity.[Pubmed: 19495827] | We have recently showed antibacterial activity against E. coli in vitro of a trademark Mega-Gluflex-containing Glucosamine sulfate (GS) and chondroitin sulfate (CS). The purpose of this study was to examine the antibacterial activity of Glucosamine sulfate as a new trademark Arthryl in vitro.
METHODS AND RESULTS:
We used cabbage and chicken broths and milk (every media of 20 ml) left opened for 1 week with and without Arthryl supplements 1,500 mg, the content of one package of the medication. A similar volume (20 ml) is ingested in taking the medication. Experiments with three repeatable results were taken for consideration. Arthryl inhibited environmental bacterial colonies' growth in every media but fungi growth was not impaired. Milk stayed liquid for the whole week with supplement of the Arthryl compared with sour milk transformation without Arthryl. Sample B showed inhibitory properties of the bacterial colonies on the fungi growth. The sample with Arthryl showed progressive growth of fungi without bacterial growth after 10 days of follow up compared with bacterial growth on media without Arthryl. Glucosamine sulfate as a new trademark
CONCLUSIONS:
Arthryl has environmental antibacterial properties but does not inhibit growth of fungal colonies. |
|
| In vivo: |
| Osteoarthritis Cartilage. 1994 Mar;2(1):61-9. | | Glucosamine sulfate compared to ibuprofen in osteoarthritis of the knee.[Pubmed: 11548225] | Glucosamine sulfate is able to stimulate proteoglycan synthesis by chondrocytes and has mild anti-inflammatory properties. In clinical trials, Glucosamine sulfate was more effective than placebo in controlling the symptoms of osteoarthritis (OA).
METHODS AND RESULTS:
In order to better characterize this therapeutic activity, we conducted a randomized, double-blind, parallel-group study of Glucosamine sulfate 500 mg t.i.d. vs ibuprofen 400 mg t.i.d., orally for 4 weeks. The study included 200 hospitalized patients with active OA of the knee, symptoms for at least 3 months and a Lequesne's index of at least 7 points. Patients were evaluated weekly. Response was defined as a reduction in the Lequesne's index by at least 2 points if the enrollment value was higher than 12 points, or by at least 1 point if the enrollment value was 12 or less points, together with a positive overall assessment by the investigator. The improvement tended to be sooner under ibuprofen (48% responders vs 28% after the 1st treatment week; P = 0.06, Fisher's Exact test), but there was no difference from the 2nd week onward, with a success rate of 52% in the ibuprofen group and of 48% in the glucosamine group (P = 0.67) at the end of treatment. The average Lequesne's index at enrollment was around 16 points and decreased by over 6 points in both groups, again with the above described trend. On the other hand, 35% of patients on ibuprofen reported adverse events, mainly of gastrointestinal origin, vs 6% adverse events with glucosamine (P < 0.001, Fisher's Exact test). The number of adverse event related drop-outs was different between the two groups (7% vs 1%, respectively; P = 0.035). Glucosamine sulfate was therefore as effective as ibuprofen on symptoms of knee OA.
CONCLUSIONS:
These data confirm Glucosamine sulfate as a safe symptomatic Slow Acting Drug for OA. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)