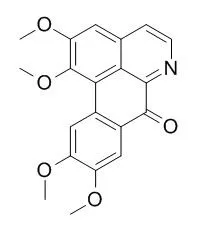
| In vitro: |
| Inorg Chem. 2012 Feb 20;51(4):1998-2009. | | TCM active ingredient oxoglaucine metal complexes: crystal structure, cytotoxicity, and interaction with DNA.[Pubmed: 22309171] | The alkaloid Oxoglaucine (OG), which is a bioactive component from traditional Chinese medicine (TCM), was synthesized by a two-step reaction and used as the ligand to react with transition metal salts to give four complexes: [OGH][AuCl(4)]·DMSO (1), [Zn(OG)(2)(H(2)O)(2)](NO(3))(2) (2), [Co(OG)(2)(H(2)O)(2)](ClO(4))(2) (3), and [Mn(OG)(2)(H(2)O)(2)](ClO(4))(2) (4).
METHODS AND RESULTS:
The crystal structures of the metal complexes were confirmed by single crystal X-ray diffraction. Complex 1 is an ionic compound consisting of a charged ligand [OGH](+) and a gold complex [AuCl(4)](-). Complexes 2-4 all have similar structures (inner-spheres), that is, octahedral geometry with two OG coordinating to one metal center and two aqua ligands occupying the two apical positions of the octahedron, and two NO(3)(-) or ClO(4)(-) as counteranions in the outer-sphere. The complexation of OG to metal ion was confirmed by ESI-MS, capillary electrophoresis and fluorescence polarization. The in vitro cytotoxicity of these complexes toward a various tumor cell lines was assayed by the MTT method. The results showed that most of these metal-Oxoglaucine complexes exhibited enhanced cytotoxicity compared with Oxoglaucine and the corresponding metal salts, with IC(50) values ranging from 1.4 to 32.7 μM for sensitive cancer cells, which clearly implied a positive synergistic effect. Moreover, these complexes appeared to be selectively active against certain cell lines.
CONCLUSIONS:
The interactions of Oxoglaucine and its metal complexes with DNA and topoisomerase I were investigated by UV-vis, fluorescence, CD spectroscopy, viscosity, and agarose gel electrophoresis, and the results indicated that these OG-metal complexes interact with DNA mainly via intercalation. Complexes 2-4 are metallointercalators, but complex 1 is not. These metal complexes could effectively inhibit topoisomerase I even at low concentration. Cell cycle analysis revealed that 1-3 caused S-phase cell arrest. | | J Nat Prod. 1998 Dec;61(12):1457-61. | | Antiplatelet aggregation constituents from Annona purpurea[Pubmed: 9868142 ] |
METHODS AND RESULTS:
Bioactivity-directed fractionation led to the isolation of 19 compounds, including three oxoaporphines, oxopurpureine (5), oxonuciferine (6), and Oxoglaucine (7); three aporphines, (+)-predicentrine (8), (-)-glaucine (9), and thalbaicalidine (10); one aporphine sensu stricto, N-formyl-purpureine (11); one proaporphine, glaziovine; one phenanthrene, thalicpureine (12); two 6a,7-dehydroaporphines, dehydrolirinidine (13) and 7-hydroxy-dehydroglaucine (14); four flavonoids, quercetin-3-O-rhamnoside, kaempferol-3-O-rhamnoside, isorhamnetin-3-O-rhamnoside, and tanarixetin-3-O-rhamnoside; one purine, adenine; one lactam amide, squamolone; and two steroids, beta-sitosterol and beta-sitosterol-beta-D-glucoside from the MeOH extract of the leaves of Formosan Annona purpurea.
CONCLUSIONS:
Among them, 11-14 were characterized as new compounds and alkaloids, 5-8, 10, and 12-14 exhibited significant antiplatelet aggregation activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)