| Structure Identification: |
| J Nat Prod. 2006 Apr;69(4):536-41. | | Serotonergic activity-guided phytochemical investigation of the roots of Angelica sinensis.[Pubmed: 16643021 ] |
METHODS AND RESULTS:
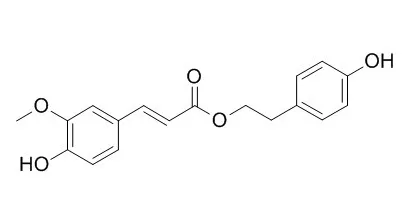
Serotonin receptor (5-HT(7)) binding assay-directed fractionation of a methanol extract of the dried roots of Angelica sinensis led to the isolation and identification of 21 compounds including a new phenolic ester, angeliferulate (1), and three new phthalides, 10-angeloylbutylphthalide (2), sinaspirolide (3), and ansaspirolide (4), along with 17 known compounds, p-Hydroxyphenethyl trans-ferulate (5), Z-ligustilide (6), Z-butylidenephthalide (7), senkyunolide I (8), Z-6-hydroxy-7-methoxydihydroligustilide (9), N-butylbenzenesulfonamide (10), 11(S),16(R)-dihydroxyoctadeca-9Z,17-diene-12,14-diyn-1-yl acetate (11), (3R,8S)-falcarindiol (12), heptadeca-1-en-9,10-epoxy-4,6-diyne-3,8-diol (13), oplopandiol (14), 8-hydroxy-1-methoxy-, Z-9-heptadecene-4,6-diyn-3-one (15), imperatorin, ferulic acid, vanillin, stigmasterol, sucrose, and 1,3-dilinolenin. This is the first report of a sulfonamide (10) identified from a higher plant source, although its presence needs further investigation. Biosynthetic pathways for dimeric phthalides 3 and 4 are proposed.
CONCLUSIONS:
Compounds p-Hydroxyphenethyl trans-ferulate, 7, 11, 12, 15, and imperatorin exhibited affinity toward 5-HT(7) receptors in a competitive binding assay. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)