| In vitro: |
| Appl Environ Microbiol. 2002 Sep;68(9):4322-7. | | Lactobacillus plantarum MiLAB 393 produces the antifungal cyclic dipeptides cyclo(L-Phe-L-Pro) and cyclo(L-Phe-trans-4-OH-L-Pro) and 3-phenyllactic acid.[Pubmed: 12200282] | We have isolated a Lactobacillus plantarum strain (MiLAB 393) from grass silage that produces broad-spectrum antifungal compounds, active against food- and feed-borne filamentous fungi and yeasts in a dual-culture agar plate assay.
METHODS AND RESULTS:
Fusarium sporotrichioides and Aspergillus fumigatus were the most sensitive among the molds, and Kluyveromyces marxianus was the most sensitive yeast species. No inhibitory activity could be detected against the mold Penicillium roqueforti or the yeast Zygosaccharomyces bailii. An isolation procedure, employing a microtiter well spore germination bioassay, was devised to isolate active compounds from culture filtrate. Cell-free supernatant was fractionated on a C(18) SPE column, and the 95% aqueous acetonitrile fraction was further separated on a preparative HPLC C(18) column. Fractions active in the bioassay were then fractionated on a porous graphitic carbon column. The structures of the antifungal compounds Cyclo(L-Phe-L-Pro), cyclo(L-Phe-trans-4-OH-L-Pro) and 3-phenyllactic acid (L/D isomer ratio, 9:1), were determined by nuclear magnetic resonance spectroscopy, mass spectrometry, and gas chromatography. MIC values against A. fumigatus and P. roqueforti were 20 mg ml(-1) for Cyclo(L-Phe-L-Pro) and 7.5 mg ml(-1) for phenyllactic acid.
CONCLUSIONS:
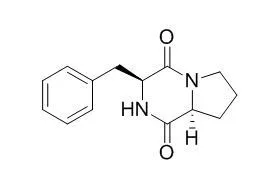
Combinations of the antifungal compounds revealed weak synergistic effects. The production of the antifungal cyclic dipeptides Cyclo(L-Phe-L-Pro) and cyclo(L-Phe-trans-4-OH-L-Pro) by lactic acid bacteria is reported here for the first time. | | J Microbiol Biotechnol. 2017 Jul 28;27(7):1249-1256. | | Isolation, Purification, and Characterization of Five Active Diketopiperazine Derivatives from Endophytic Streptomyces SUK 25 with Antimicrobial and Cytotoxic Activities.[Pubmed: 28535606] | In our search for new sources of bioactive secondary metabolites from Streptomyces sp., the ethyl acetate extracts from endophytic Streptomyces SUK 25 afforded five active diketopiperazine (DKP) compounds.
METHODS AND RESULTS:
The aim of this study was to characterize the bioactive compounds isolated from endophytic Streptomyces SUK 25 and evaluate their bioactivity against multiple drug resistance (MDR) bacteria such as Enterococcus raffinosus, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumanii, Pseudomonas aeruginosa, and Enterobacter spp., and their cytotoxic activities against the human hepatoma (HepaRG) cell line. The production of secondary metabolites by this strain was optimized through Thornton's medium. Isolation, purification, and identification of the bioactive compounds were carried out using high-performance liquid chromatography, high-resolution mass liquid chromatography-mass spectrometry, Fourier transform infrared spectroscopy, and nuclear magnetic resonance, and cryopreserved HepaRG cells were selected to test the cytotoxicity. The results showed that endophytic Streptomyces SUK 25 produces four active DKP compounds and an acetamide derivative, which were elucidated as cyclo-(L-Val-L-Pro), cyclo-(L-Leu-L-Pro), Cyclo(L-Phe-L-Pro), cyclo-(L-Val-L-Phe), and N-(7-hydroxy-6-methyl-octyl)-acetamide. These active compounds exhibited activity against methicillin-resistant S. aureus ATCC 43300 and Enterococcus raffinosus, with low toxicity against human hepatoma HepaRG cells.
CONCLUSIONS:
Endophytic Streptomyces SUK 25 has the ability to produce DKP derivatives biologically active against some MDR bacteria with relatively low toxicity against HepaRG cells line. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)