| In vitro: |
| Int J Mol Sci . 2021 Feb 5;22(4):1610. | | Diphlorethohydroxycarmalol Isolated from Ishige okamurae Exerts Vasodilatory Effects via Calcium Signaling and PI3K/Akt/eNOS Pathway[Pubmed: 33562632] | | Nitric oxide (NO) is released by endothelial cells in the blood vessel wall to enhance vasodilation. Marine polyphenols are known to have protective effects against vascular dysfunction and hypertension. The present study is the first to investigate how Diphlorethohydroxycarmalol (DPHC) isolated from Ishige okamurae affects calcium levels, resulting in enhanced vasodilation. We examined calcium modulation with the well-known receptors, acetylcholine receptor (AchR) and vascular endothelial growth factor 2 (VEGFR2), which are related to NO formation, and further confirmed the vasodilatory effect of DPHC. We confirmed that DPHC stimulated NO production by increasing calcium levels and endothelial nitric oxide synthase (eNOS) expression. DPHC affected AchR and VEGFR2 expression, thereby influencing transient calcium intake. Specific antagonists, atropine and SU5416, were used to verify our findings. Furthermore, based on the results of in vivo experiments, we treated Tg(flk:EGFP) transgenic zebrafish with DPHC to confirm its vasodilatory effect. In conclusion, the present study showed that DPHC modulated calcium transit through AchR and VEGFR2, increasing endothelial-dependent NO production. Thus, DPHC, a natural marine component, can efficiently ameliorate cardiovascular diseases by improving vascular function. | | Cell Biochem Funct . 2021 Jun;39(4):546-554. | | Diphlorethohydroxycarmalol inhibits melanogenesis via protein kinase A/cAMP response element-binding protein and extracellular signal-regulated kinase-mediated microphthalmia-associated transcription factor downregulation in α-melanocyte stimulating hormone-stimulated B16F10 melanoma cells and zebrafish[Pubmed: 33474761] | | Diphlorethohydroxycarmalol (DPHC) is a marine polyphenolic compound derived from brown alga Ishige okamurae. A previously study has suggested that DPHC possesses strong mushroom tyrosinase inhibitory activity. However, the anti-melanogenesis effect of DPHC has not been reported at cellular level. The objective of the present study was to clarify the melanogenesis inhibitory effect of DPHC and its molecular mechanisms in murine melanoma cells (B16F10) and zebrafish model. DPHC significantly inhibited tyrosinase activity and melanin content dose-dependently in α-melanocyte stimulating hormone (α-MSH)-stimulated B16F10 cells. This polyphenolic compound also suppressed the expression of phosphorylation of cAMP response element-binding protein (CREB) by attenuating phosphorylation of cAMP-dependent protein kinase A, resulting in decreased MITF expression levels. Furthermore, DPHC downregulated MITF protein expression levels by promoting the phosphorylation of extracellular signal-regulated kinase. It also inhibited tyrosinase, tyrosinase-related protein 1 (TRP-1), and TRP-2 in α-MSH stimulated B16F10 cells. In in vivo studies using zebrafish, DPHC also markedly inhibited melanin synthesis in a dose-dependent manner. These results demonstrate that DPHC can effectively inhibit melanogenesis in melanoma cells in vitro and in zebrafish in vivo, suggesting that DPHC could be applied in fields of pharmaceutical and cosmeceuticals as a skin-whitening agent. Significance of study: The present study showed for the first time that DPHC could inhibit a-MSH-stimulated melanogenesis via PKA/CREB and ERK pathway in melanoma cells. It also could inhibit pigmentation in vivo in a zebrafish model. This evidence suggests that DPHC has potential as a skin whitening agent. Taken together, DPHC could be considered as a novel anti-melanogenic agent to be applied in cosmetic, food, and medical industry. | | Mar Drugs . 2015 Apr 13;13(4):2141-57. | | Diphlorethohydroxycarmalol inhibits interleukin-6 production by regulating NF-κB, STAT5 and SOCS1 in lipopolysaccharide-stimulated RAW264.7 cells[Pubmed: 25871292] | | Diphlorethohydroxycarmalol (DPHC) is a phlorotannin compound isolated from Ishige okamuarae, a brown alga. This study was conducted to investigate the anti-inflammatory effect and action mechanism of DPHC in lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophages. We found that DPHC strongly reduces the production of interleukin 6 (IL-6), but not that of tumor necrosis factor-alpha (TNF-α) induced by LPS. DPHC (12.5 and 100 μM) suppressed the phosphorylation and the nuclear translocation of NF-kappaB (NF-κB), a central signaling molecule in the inflammation process induced by LPS. The suppressor of cytokine signaling 1 (SOCS1) is a negative feedback regulator of Janus kinase (Jak)-signal transducer and activator of transcription (STAT) signaling. In this study, DPHC inhibited STAT5 expression and upregulated that of SOCS1 at a concentration of 100 μM. Furthermore, N-tosyl-l-phenylalanine chloromethyl ketone (TPCK) (a specific NF-κB inhibitor) and JI (a specific Jak2 inhibitor) reduced the production of IL-6, but not that of tumor necrosis factor-alpha (TNF-α) in LPS-stimulated RAW 264.7 macrophages. These findings demonstrate that DPHC inhibits IL-6 production via the downregulation of NF-κB and Jak2-STAT5 pathway and upregulation of SOCS1. | | Mar Drugs . 2020 Oct 26;18(11):529. | | Diphlorethohydroxycarmalol (DPHC) Isolated from the Brown Alga Ishige okamurae Acts on Inflammatory Myopathy as an Inhibitory Agent of TNF-α[Pubmed: 33114618] | | Inflammation affects various organs of the human body, including skeletal muscle. Phlorotannins are natural biologically active substances found in marine brown algae and exhibit anti-inflammatory activities. In this study, we focused on the effects of phlorotannins on anti-inflammatory activity and skeletal muscle cell proliferation activity to identify the protective effects on the inflammatory myopathy. First, the five species of marine brown algal extracts dramatically inhibited nitric oxide (NO) production in lipopolysaccharide (LPS)-induced RAW 264.7 cells without toxicity at all the concentrations tested. Moreover, the extracts collected from Ishige okamurae (I. okamurae) significantly increased cell proliferation of C2C12 myoblasts compared to the non-treated cells with non-toxicity. In addition, as a result of finding a potential tumor necrosis factor (TNF)-α inhibitor that regulates the signaling pathway of muscle degradation in I. okamurae-derived natural bioactive compounds, Diphlorethohydroxycarmalol (DPHC) is favorably docked to the TNF-α with the lowest binding energy and docking interaction energy value. Moreover, DPHC down-regulated the mRNA expression level of pro-inflammatory cytokines and suppressed the muscle RING-finger protein (MuRF)-1 and Muscle Atrophy F-box (MAFbx)/Atrgoin-1, which are the key protein muscle atrophy via nuclear factor-κB (NF-κB), and mitogen-activated protein kinase (MAPKs) signaling pathways in TNF-α-stimulated C2C12 myotubes. Therefore, it is expected that DPHC isolated from IO would be developed as a TNF-α inhibitor against inflammatory myopathy. |
|


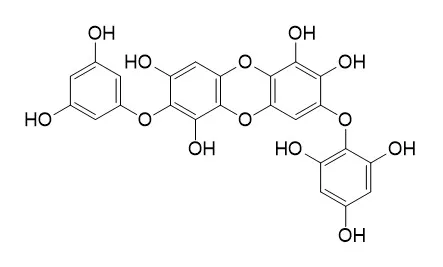



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)