| In vitro: |
| Biochem Biophys Res Commun. 2014 Jul 4;449(3):307-12. | | Antiviral effects of two Ganoderma lucidum triterpenoids against enterovirus 71 infection.[Pubmed: 24845570] | Enterovirus 71 (EV71) is a major causative agent for hand, foot and mouth disease (HFMD), and fatal neurological and systemic complications in children. However, there is currently no clinical approved antiviral drug available for the prevention and treatment of the viral infection.
METHODS AND RESULTS:
Here, we evaluated the antiviral activities of two Ganoderma lucidum triterpenoids (GLTs), Lanosta-7,9(11),24-trien-3-one,15;26-dihydroxy (GLTA) and Ganoderic acid Y (GLTB), against EV71 infection. The results showed that the two natural compounds display significant anti-EV71 activities without cytotoxicity in human rhabdomyosarcoma (RD) cells as evaluated by 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell proliferation assay. The mechanisms by which the two compounds affect EV71 infection were further elucidated by three action modes using Ribavirin, a common antiviral drug, as a positive control.
CONCLUSIONS:
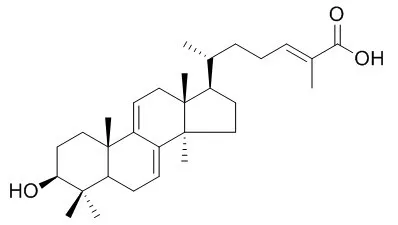
The results suggested that GLTA and GLTB prevent EV71 infection through interacting with the viral particle to block the adsorption of virus to the cells. In addition, the interactions between EV71 virion and the compounds were predicated by computer molecular docking, which illustrated that GLTA and GLTB may bind to the viral capsid protein at a hydrophobic pocket (F site), and thus may block uncoating of EV71. Moreover, we demonstrated that GLTA and GLTB significantly inhibit the replication of the viral RNA (vRNA) of EV71 replication through blocking EV71 uncoating. Thus, GLTA and GLTB may represent two potential therapeutic agents to control and treat EV71 infection. | | Appl Environ Microbiol. 2005 Jul;71(7):3653-8. | | Effect of 26-oxygenosterols from Ganoderma lucidum and their activity as cholesterol synthesis inhibitors.[Pubmed: 16000773] | Ganoderma lucidum is a medicinal fungus belonging to the Polyporaceae family which has long been known in Japan as Reishi and has been used extensively in traditional Chinese medicine.
METHODS AND RESULTS:
We report the isolation and identification of the 26-oxygenosterols ganoderol A, ganoderol B, ganoderal A, and Ganoderic acid Y and their biological effects on cholesterol synthesis in a human hepatic cell line in vitro. | | Phytochemistry . 2018 May;149:103-115. | | Lanostane triterpenes from the mushroom Ganoderma resinaceum and their inhibitory activities against α-glucosidase[Pubmed: 29490285] | | Abstract
Eighteen previously undescribed lanostane triterpenes and thirty known analogues were obtained from the fruiting bodies of Ganoderma resinaceum. Resinacein C was isolated from a natural source for the first time. The structures of all the above compounds were elucidated by extensive spectroscopic analysis and comparisons of their spectroscopic data with those reported in the literature. Furthermore, in an in vitro assay, Resinacein C, Ganoderic acid Y, lucialdehyde C, 7-oxo-ganoderic acid Z3, 7-oxo-ganoderic acid Z, and lucidadiol showed strong inhibitory effects against α-glucosidase compared with the positive control drug acarbose. The structure-activity relationships of ganoderma triterpenes on α-glucosidase inhibition showed that the C-24/C-25 double bond is necessary for α-glucosidase inhibitory activity. Moreover, the carboxylic acid group at C-26 and the hydroxy group at C-15 play important roles in enhancing inhibitory effects of these triterpenes.
Keywords: Ganoderma resinaceum; Ganodermataceae; Lanostane triterpenes; α-Glucosidase inhibitor. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)