| In vitro: |
| J Nat Prod. 2015 Oct 23;78(10):2481-7 | | Chalcones from Angelica keiskei: Evaluation of Their Heat Shock Protein Inducing Activities.[Pubmed: 26431394] |
METHODS AND RESULTS:
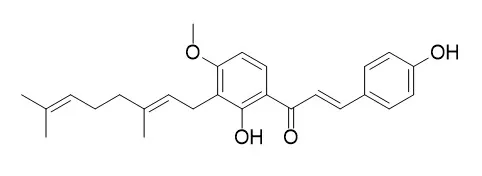
Five new chalcones, 4,2',4'-trihydroxy-3'-[(2E,5E)-7-methoxy-3,7-dimethyl-2,5-octadienyl]chalcone (1), (±)-4,2',4'-trihydroxy-3'-[(2E)-6-hydroxy-7-methoxy-3,7-dimethyl-2-octenyl]chalcone (2), 4,2',4'-trihydroxy-3'-[(2E)-3-methyl-5-(1,3-dioxolan-2-yl)-2-pentenyl]chalcone (3), 2',3'-furano-4-hydroxy-4'-methoxychalcone (4), and (±)-4-hydroxy-2',3'-(2,3-dihydro-2-methoxyfurano)-4'-methoxychalcone (5), were isolated from the aerial parts of Angelica keiskei Koidzumi together with eight known chalcones, 6-13, which were identified as (±)-4,2',4'-trihydroxy-3'-[(6E)-2-hydroxy-7-methyl-3-methylene-6-octenyl]chalcone (6), xanthoangelol (7), Xanthoangelol F (8), xanthoangelol G (9), 4-hydroxyderricin (10), xanthoangelol D (11), xanthoangelol E (12), and xanthoangelol H (13), respectively. Chalcones 1-13 were evaluated for their promoter activity on heat shock protein 25 (hsp25, murine form of human hsp27). Compounds 1 and 6 activated the hsp25 promoter by 21.9- and 29.2-fold of untreated control at 10 μM, respectively. Further protein expression patterns of heat shock factor 1 (HSF1), HSP70, and HSP27 by 1 and 6 were examined. Compound 6 increased the expression of HSF1, HSP70, and HSP27 by 4.3-, 1.5-, and 4.6-fold of untreated control, respectively, without any significant cellular cytotoxicities, whereas 1 did not induce any expression of these proteins.
CONCLUSIONS:
As a result, 6 seems to be a prospective HSP inducer. | | Planta Med. 2001 Apr;67(3):230-5. | | Artery relaxation by chalcones isolated from the roots of Angelica keiskei.[Pubmed: 11345693 ] | An EtOAc-soluble fraction from a 50% EtOH extract of the roots of Angelica keiskei inhibited phenylephrine-induced vasoconstriction in rat aortic rings, while an EtOAc-insoluble fraction had no effect at 100 micrograms/ml.
METHODS AND RESULTS:
Five active substances isolated from the EtOAc-soluble fraction of the roots were identified as xanthoangelol (1), 4-hydroxyderricin (2), and xanthoangelols B (3), E (4) and F (5), which inhibited phenylephrine-induced vasoconstriction at the concentrations of 10-100 micrograms/ml. It was found that xanthoangelol (1), 4-hydroxyderricin (2), and xanthoangelol E (4) and Xanthoangelol F (5) inhibited the phenylephrine-induced vasoconstriction through endothelium-dependent endothelium-derived relaxing factor (EDRF) production and/or nitric oxide (NO) production. Among the five chalcones, xanthoangelol B (3) inhibited the phenylephrine-induced vasoconstriction most strongly, and it inhibited the phenylephrine-induced vasoconstriction in the presence or absence of endothelium and in the presence or absence of NG-monomethyl-L-arginine (L-NMMA) (an NO synthetase inhibitor). Furthermore, 4-hydroxyderricin (2) and xanthoangelol B (3) at concentrations of 10-100 micrograms/ml concentration-dependently inhibited the elevation of intracellular free calcium [Ca2+]i induced by phenylephrine.
CONCLUSIONS:
These results demonstrate that compounds 1, 2, 4 and 5 inhibit phenylephrine-induced vasoconstriction through endothelium-dependent production of EDRF/NO and/or through the reduction of the [Ca2+]i elevation induced by phenylephrine. On the other hand, the inhibitory mechanism of compound 3 on phenylephrine-induced vasoconstriction might involve the direct inhibition of smooth muscle functions through the reduction of [Ca2+]i elevation without affecting EDRF/NO production. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)