| Description: |
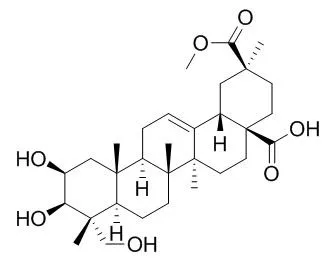
Phytolaccagenin has promising antifungal activity against ATCC standard cultures of Candida albicans and Cryptococcus neoformans, and against clinical isolates of these fungi, it also shows inhibitory effects on lipopolysaccharide-induced NO production, and haemolytic activities. |
| Targets: |
Antifection | NO |
| In vitro: |
| Nat. Prod. Commun.,2010, 5(7):1013-8. | | Antifungal activity of saponin-rich extracts of Phytolacca dioica and of the sapogenins obtained through hydrolysis.[Pubmed: 20734930] |
METHODS AND RESULTS:
A saponin-rich extract of Phytolacca dioica L. berries, its acid hydrolysate, and its major aglycone, Phytolaccagenin, were assayed for antifungal activity against ATCC standard cultures of Candida albicans and Cryptococcus neoformans, and against clinical isolates of these fungi.
The activity of the extract was either low or negligible, but the hydrolysate, containing the sapogenins, including Phytolaccagenin, and also pure Phytolaccagenin, showed promising antifungal potency.
CONCLUSIONS:
Hydrolysis of a natural product extract is shown to be a useful modification leading to improved bioactivity. |
|
| In vivo: |
| J Pharm Biomed Anal . 2015 Mar 25;107:82-8. | | Development and validation of a HPLC-MS/MS method for the determination of phytolaccagenin in rat plasma and application to a pharmacokinetic study[Pubmed: 25575173] | | Abstract
Radix Phytolaccae (the dried root of Phytolacca acinosa Roxb. or Phytolacca americana L.) is widely used in East Asian countries for the treatment of inflammation-related diseases. The active component of Radix Phtolaccae is Phytolcaccagenin a triterpenoid saponin. Phytolcaccagenin has anti-inflammatory activities that exceed those of Esculentoside A and its derivatives regarding suppression of LPS-induced inflammation, and has a lower toxicity profile with less hemolysis. To date, no information is available about analytical method and pharmacokinetic studies of Phytolaccagenin. To explore PK profile of this compound, a HPLC-MS/MS assay of Phytolaccagenin in rat plasma was developed and validated. The method was fully validated according to FDA Guidance for industry. The detection was performed by a triple-quadrupole tandem mass spectrometer with multiple reactions monitoring (MRM) in positive ion mode via electrospray ionization. The monitored transitions were m/z 533.2>515.3 for Phytolcaccagenin, and 491.2>473.2 for I.S. The analysis was performed on a Symmetry C18 column (4.6 mm × 50 mm, 3.5 μm) using gradient elution with the mobile phase consisting of acetonitrile and 0.1% formic acid water at a flow rate of 1 ml/min with a 1:1 splitter ratio. The method was validated with a LLOQ of 20 ng/ml and an ULOQ of 1000 ng/ml. The response versus concentration data were fitted with 1/x weighting and the correlation coefficient (r) were greater than 0.999. The average matrix effect and the average extraction recovery were acceptable. This validation in rat plasma demonstrated that Phytolaccagenin was stable for 30 days when stored below -20°C, for 6h at room temperature (RT, 22°C), for 12 h at RT for prepared control samples in auto-sampler vials, and during three successive freeze/thaw cycles results at -20°C. The validated method has been successfully applied to an intravenous bolus pharmacokinetic study of Phytolaccagenin in male Sprague-Dawley rats (10 mg/kg, i.v.). Blood samples taken from 0 to 24h after injection were collected, and data analyzed with WinNonlin. The half-life and clearance were 1.4±0.9 h and 2.1±1.1 L/h/kg, respectively.
Keywords: Anti-inflammatory; HPLC–MS/MS; Pharmacokinetic study; Phytolaccagenin; Validation. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)