| In vitro: |
| J Biol Chem. 1981 May 10;256(9):4253-8. | | The in vitro biosynthesis of taxiphyllin and the channeling of intermediates in Triglochin maritima.[Pubmed: 7012151] |
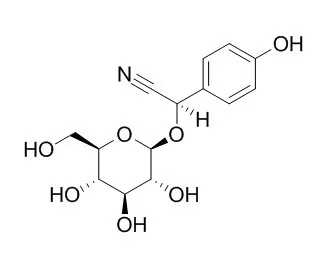
The in vitro biosynthesis of the cyanogenic glucoside Taxiphyllin has recently been demonstrated in Triglochin maritima (Hösel, W., and Nahrstedt, A. (1980) Arch. Biochem. Biophys. 203, 753-757).
METHODS AND RESULTS:
We have now studied in more detail the multistep conversion of tyrosine into p-hydroxymandelonitrile, the immediate precursor of Taxiphyllin, catalyzed by microsomes isolated from dark-grown seedlings. The biosynthetic pathway involves N-hydroxytyrosine, p-hydroxyphenylacetaldoxime, and p-hydroxyphenylacetonitrile. In marked contrast to an analogous pathway in Sorghum bicolor, p-hydroxyphenylacetonitrile is the best substrate for cyanide production (Vmax = 224 nmol/h/g, fresh wt) and the physiological substrate tyrosine is the poorest (Vmax = 18.8 nmol/h/g, fresh wt). The substrates exhibit alkaline pH optima between 7.5 and 9, and all except tyrosine show pronounced substrate inhibition. We have found that p-hydroxyphenylacetonitrile generated in situ from tyrosine is free to equilibrate by diffusion with exogenous material. On the other hand, neither N-hydroxytyrosine nor p-hydroxyphenylacetaldoxime will readily exchange with exogenous intermediates.
CONCLUSIONS:
We consider both N-hydroxytyrosine and p-hydroxyphenylacetaldoxime to be channeled in T. maritima, whereas in S. bicolor N-hydroxytyrosine and p-hydroxyphenylacetonitrile are channeled and the aldoxime is freely exchangeable. | | J. Nat. Prod.,2006:69(9):1366-9. | | Phytochemical constituents from Salsola tetrandra.[Pubmed: 16989538 ] |
METHODS AND RESULTS:
The new norisoprenoid 3beta-hydroxy-5alpha,6alpha-epoxy-beta-ionone-2alpha-O-beta-d-glucopyranoside (1) and the long-chain hydroxy fatty acids 9,12,13-trihydroxyoctadeca-10(E),15(Z)-dienoic acid (2) and 9,12,13-trihydroxyoctadeca-10(E)-dienoic acid (3) were isolated from Salsola tetrandra aerial parts, together with 3,4,5-trimethoxyphenyl-beta-d-glucopyranoside (4), 9-hydroxylinaloyl glucoside (5), Taxiphyllin (6), trans-N-feruloyltyramine (7), and S-(-)-trans-N-feruloyloctopamine (8). Their structures were elucidated by extensive spectroscopic analysis and chemical methods.
CONCLUSIONS:
Compounds 6 and 8 displayed mild antibacterial activity against Staphylococcus aureus, whereas compound 6 showed the highest activity in the Artemia salina bioassay. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)